NAPOLI-3 Trial: A Breakthrough in Metastatic Pancreatic Cancer Treatment
Comprehensive review of NAPOLI 3 trials results
NAPOLI-3
Trial: A Breakthrough in Metastatic Pancreatic Cancer Treatment
Article Citation
Title: NALIRIFOX versus nab-paclitaxel and gemcitabine in treatment-naive
patients with metastatic pancreatic ductal adenocarcinoma (NAPOLI 3): a
randomised, open-label, phase 3 trial
Authors: Zev A Wainberg, Davide Melisi, Teresa Macarulla, Roberto Pazo Cid,
Sreenivasa R Chandana, and colleagues
Journal: The Lancet
Publication Date: September 11, 2023 (online); October 7, 2023 (print)
DOI: 10.1016/S0140-6736(23)01366-1
Trial Registration: NCT04083235
🎯 5 Key Clinical Takeaways
1. First
Head-to-Head Chemotherapy Comparison Establishes NALIRIFOX Superiority
NAPOLI-3 is the landmark phase 3 trial directly comparing two
combination chemotherapy regimens in treatment-naive metastatic pancreatic
cancer. NALIRIFOX achieved median overall survival of 11.1 months versus 9.2
months with nab-paclitaxel plus gemcitabine (HR 0.83, p=0.036),
representing a 17% reduction in death risk.
2. Unprecedented
Long-Term Survival Gains
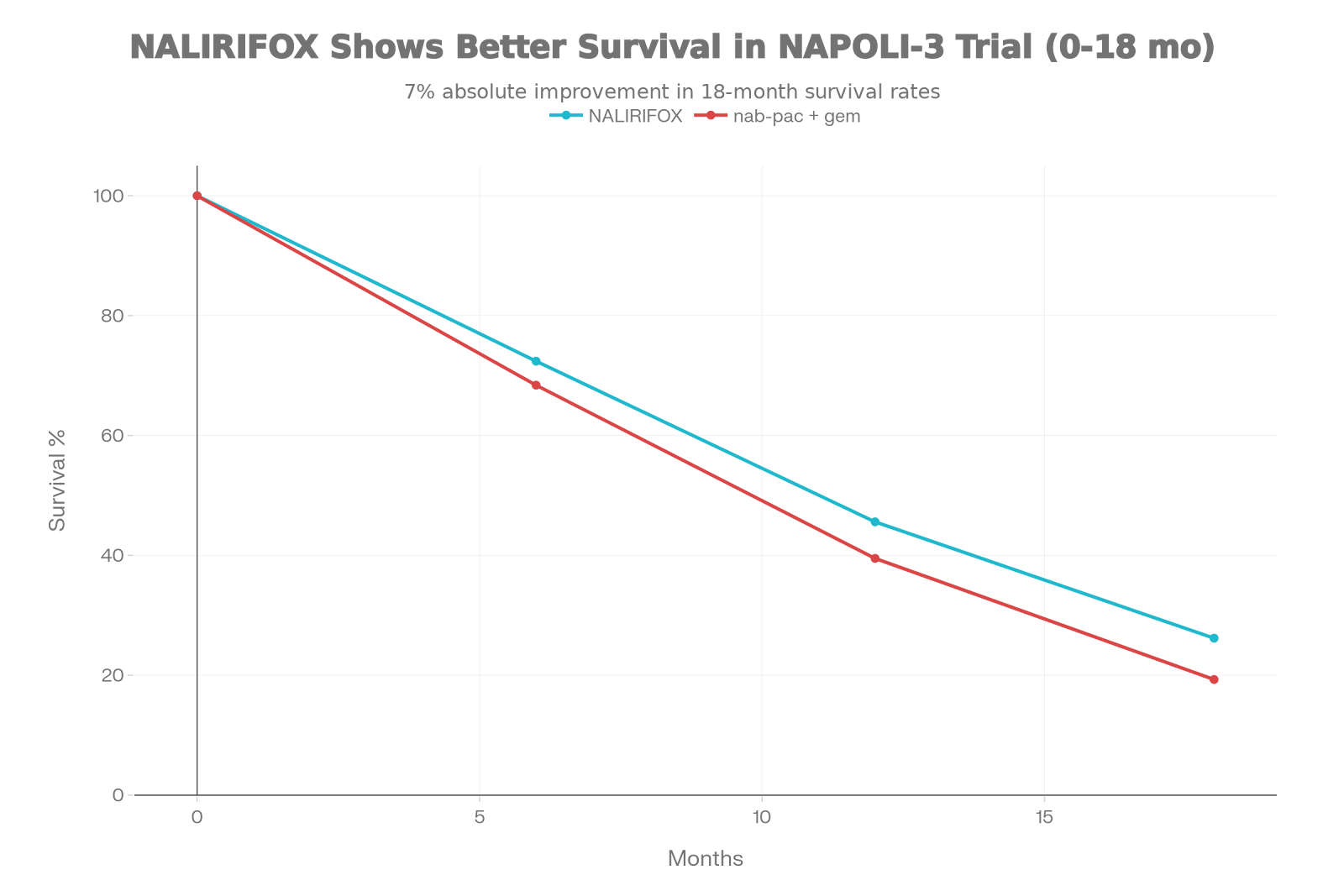
The survival advantage persists over time, with NALIRIFOX achieving:
– 18-month OS: 26.2% vs 19.3% (36% relative increase in long-term
survivors) – 12-month OS: 45.6% vs 39.5%
This durable benefit contrasts with therapies showing early
separation that converges over time, suggesting sustained therapeutic efficacy.
3. Paradoxically
Superior Safety Profile Despite Quadruplet Therapy
Despite combining four chemotherapy agents, NALIRIFOX
demonstrated: – Lower neutropenia: 14% vs 25% (grade 3-4) – Lower
anemia: 11% vs 17% – Reduced peripheral neuropathy: 3% vs 6%
The liposomal irinotecan formulation and reduced oxaliplatin dosing
enable enhanced tumor penetration while minimizing systemic toxicity.
4. Doubled
Progression-Free Survival at 12 Months
NALIRIFOX extended median PFS to 7.4 months versus 5.6 months
(HR 0.69, p<0.0001), with 12-month PFS nearly doubling (27.4% vs
13.9%), providing patients extended disease-free intervals before progression.
5. FDA/EMA
Approval Establishes New Treatment Standard
Following NAPOLI-3, NALIRIFOX received regulatory approval in early
2024, becoming the first new first-line regimen for metastatic pancreatic
cancer approved since 2013—marking an 11-year therapeutic advancement.
📋
Clinical Context: The Pancreatic Cancer Urgency
The Devastating Disease
Landscape
Pancreatic
ductal adenocarcinoma (PDAC) remains one of oncology’s most lethal
malignancies:
•
5-year survival for
metastatic disease: Only 3%
•
Annual incidence: ~60,000 USA; ~500,000 globally
•
Median survival without
treatment: <6 months
Over
the past decade, two combination chemotherapy regimens emerged as first-line
standards:
|
Regimen |
Key |
Year |
Median |
Comparator |
|
FOLFIRINOX |
PRODIGE |
2011 |
11.1 |
Gemcitabine |
|
Nab-paclitaxel/Gemcitabine |
MPACT |
2013 |
8.5 |
Gemcitabine |
The
Critical Gap: Neither trial compared these regimens
directly against each other, leaving clinicians without definitive
evidence to guide treatment selection in daily practice.
Why NAPOLI-3 Matters
For over a decade,
treatment decisions relied on: – Indirect cross-trial comparisons –
Institutional experience and preference – Toxicity profile assumptions –
Patient performance status judgment
NAPOLI-3
eliminates this uncertainty by directly randomizing
770 comparable patients to two active regimens under identical trial
conditions, establishing definitive comparative efficacy.
🔬 Study Design:
Methodology Overview
Trial Structure
•
Type: Randomized, open-label, phase 3
•
Sites: 187 centers across 18 countries (Europe, North America, South
America, Asia, Australia)
•
Population: 770 patients (383 NALIRIFOX, 387 control)
•
Enrollment Period: February 2020 – August 2021
•
Follow-up: Median 16.1 months (IQR 13.4-19.1)
Randomization Strategy
Balanced block methodology via
interactive web/voice response system, stratified by: 1. Geographic region
(North America vs East Asia vs rest of world) 2. ECOG performance status (0 vs
1) 3. Liver metastases presence (yes vs no)
Treatment Regimens
NALIRIFOX Arm
(n=383)
•
Liposomal irinotecan: 50 mg/m²
•
Oxaliplatin: 60 mg/m² (reduced dose)
•
Leucovorin: 400 mg/m²
•
Fluorouracil: 2400 mg/m² continuous infusion
•
Schedule: Days 1 and 15 of 28-day cycles
Key Innovation: Liposomal formulation enhances tumor penetration
while minimizing bone marrow toxicity
Control Arm
(n=387)
•
Nab-paclitaxel: 125 mg/m²
•
Gemcitabine: 1000 mg/m²
•
Schedule: Days 1, 8, and 15 of 28-day cycles
📊 Study
Endpoints & Statistical Design
Primary Endpoint:
Overall Survival
•
Definition: Time from randomization to death from any cause
•
Power: 90% power to detect HR 0.75 at alpha 0.05 (two-sided)
•
Required events: 543 OS events (final analysis: 544 events)
Secondary Endpoints
1.
Progression-free survival (PFS)
by RECIST 1.1
2.
Overall response rate (ORR)
Statistical Approach
Hierarchical testing controlled type I error: – If OS significant → test PFS – If PFS
significant → test ORR
👥 Patient
Population: Who Was Enrolled?
Baseline Characteristics
|
Characteristic |
NALIRIFOX |
Control |
|
Median age |
64 (20-85) |
65 (36-82) |
|
Male (%) |
53% |
59% |
|
White (%) |
82% |
84% |
|
ECOG 0 (%) |
42% |
43% |
|
Liver |
80% |
80% |
|
Median CA |
1,856 |
1,544 |
|
Elevated CA |
84% |
82% |
Inclusion Criteria
✓ Treatment-naive
metastatic PDAC
✓ Histologically or
cytologically confirmed diagnosis
✓ ECOG performance
status 0-1
✓ ≥1 measurable lesion
per RECIST 1.1
✓ HER2-negative
disease
Exclusion Criteria
✗ Prior anticancer
therapy (except adjuvant therapy >12 months prior)
✗ Symptomatic brain
metastases
✗ Active secondary
malignancy
✗ Significant
renal/hepatic impairment
Real-World
Applicability Assessment
Strengths: –
✅ No
upper age limit (enrolled patients to age 85)
✅
Global recruitment from 187 sites (academic and community)
✅
Pragmatic eligibility criteria
✅
Median 3.0-3.6 weeks from diagnosis to randomization (treatment-naive
verification)
Limitations:
⚠️
Predominantly Caucasian (82-84%) – limited diversity
⚠️
ECOG 0-1 only – excludes frailer patients
⚠️ Requires measurable disease – excludes
isolated peritoneal disease
For
UK NHS Practice: The trial’s age distribution,
performance status mix, and metastatic burden closely mirror UK pancreatic
cancer populations, supporting strong clinical translation.
🎯 Key Efficacy Results
PRIMARY OUTCOME: Overall
Survival
Median Overall Survival:
NALIRIFOX: 11.1 months (95% CI
10.0–12.1)
Nab-paclitaxel/Gemcitabine: 9.2 months
(95% CI 8.3–10.6)
Hazard Ratio: 0.83 (95% CI 0.70–0.99, p=0.036)
Clinical Translation: 17% reduction in
death risk; 1.9-month absolute median OS improvement
Survival Curves
Show Sustained Benefit
|
Timepoint |
NALIRIFOX |
Control |
Difference |
|
6-month OS |
72.4% |
68.4% |
+4.0% |
|
12-month OS |
45.6% |
39.5% |
+6.1% |
|
18-month OS |
26.2% |
19.3% |
+6.9% ⭐ |
The 18-month OS difference represents a 36% relative increase in
long-term survivors, indicating durable—not transient—therapeutic benefit.
Sensitivity
Analysis (Censoring at Subsequent Therapy)
Controlling for confounding by post-progression treatments revealed even
greater benefit: – Median OS: 15.1 months (NALIRIFOX) vs 9.2 months
(control) – HR: 0.71 (95% CI 0.56–0.90, nominal p=0.0048)
This analysis suggests the primary ITT analysis may underestimate
true treatment effect.
SECONDARY
OUTCOME: Progression-Free Survival
Median PFS: – NALIRIFOX: 7.4
months (95% CI 6.0–7.7)
Nab-paclitaxel/Gemcitabine: 5.6 months
(95% CI 5.3–5.8)
Hazard Ratio: 0.69 (95% CI 0.58–0.83, p<0.0001)
Clinical Impact: 31% reduction in
progression/death; extended disease control interval
PFS Rates at Key Timepoints
|
Timepoint |
NALIRIFOX |
Control |
Relative Increase |
|
6-month PFS |
56.4% |
43.2% |
+30% |
|
12-month PFS |
27.4% |
13.9% |
+97% (nearly doubled) ⭐ |
|
18-month PFS |
11.4% |
3.6% |
+216% (tripled) |
The 12-month doubling of PFS (27.4% vs
13.9%) represents a clinically meaningful increase in prolonged disease
control, translating to extended time without new symptoms or need for salvage
therapy.
Overall Response Rate
& Duration
|
Metric |
NALIRIFOX |
Control |
P-value |
|
ORR (%) |
41.8 |
36.2 |
0.11 (NS) |
|
Median Duration of Response |
7.3 mo |
5.0 mo |
0.02 ⭐ |
Pattern: Similar response rates but
significantly longer response duration with NALIRIFOX, indicating
deeper, more durable disease control among responders.
Subgroup Consistency
Treatment benefits remained consistent across clinically important
subgroups:
✅ ECOG performance status (0 vs 1)
✅ Geographic region
✅ Liver metastases presence (yes vs no)
This consistency strengthens confidence that treatment benefit
applies broadly across patient populations.
⚠️ Safety
Profile: A Paradox of Quadruplet Therapy
Overall Toxicity
Burden: Similar Despite Increased Complexity
|
Safety Measure |
NALIRIFOX |
Control |
|
Any TEAE (%) |
>99 |
>99 |
|
Grade ≥3 TEAE (%) |
87 |
86 |
|
Treatment-Related Deaths (%) |
2 |
2 |
Key Finding: Despite combining four
agents, NALIRIFOX showed similar overall severe toxicity rates to doublet
therapy—a remarkable finding challenging assumptions about treatment intensity.
DISTINCT TOXICITY PROFILES
NALIRIFOX-Predominant
Toxicities (Gastrointestinal)
|
Adverse Event |
NALIRIFOX |
Control |
Clinical |
|
Diarrhea |
20% |
5% |
Acute; reversible |
|
Nausea |
12% |
3% |
Manageable with |
|
Vomiting |
7% |
2% |
Secondary to GI |
|
Hypokalemia |
15% |
4% |
Secondary to |
|
Decreased |
9% |
3% |
— |
Control
Arm-Predominant Toxicities (Hematological)
|
Adverse Event |
NALIRIFOX |
Control |
Clinical |
|
Neutropenia |
14% |
25% |
44% reduction; |
|
Anemia |
11% |
17% |
35% reduction; |
|
Peripheral |
3% |
6% |
50% reduction; |
Clinical Safety
Interpretation
Why
Lower Hematological Toxicity Despite Quadruplet Therapy?
1.
Liposomal irinotecan
formulation: Enhances tumor delivery while
minimizing bone marrow exposure
2.
Reduced oxaliplatin dose: NALIRIFOX uses 60 mg/m² vs FOLFIRINOX’s 85 mg/m²
3.
Optimized drug sequencing: Staggered administration reduces peak systemic concentrations
Treatment Duration
& Dose Modifications
|
Metric |
NALIRIFOX |
Control |
|
Median treatment duration |
24.3 |
17.6 |
|
Duration advantage |
+6.7 |
— |
|
Dose reductions needed (%) |
60% |
54% |
|
Discontinued due to AE (%) |
32% |
30% |
Interpretation: Longer treatment duration with NALIRIFOX reflects better
tolerability enabling continued therapy without excess cumulative toxicity.
Adverse Event
Management Strategies
For
Diarrhea (Most Frequent NALIRIFOX Toxicity):
–
Prophylactic loperamide starting day 1
–
Patient education on dietary modifications
–
Close monitoring weeks 1-4
–
Temporary dose holds for grade 3 events – IV fluids if dehydration develops
For
Hypokalemia:
–
Weekly electrolyte monitoring
–
Oral potassium supplementation
– IV
potassium repletion if severe (<3.0 mmol/L)
For
Nausea/Vomiting:
– NK1
antagonist prophylaxis
–
5-HT3 antagonist + dexamethasone
–
Patient-specific anti-emetic optimization
🏥
Clinical Relevance & Practice-Changing Impact
How NAPOLI-3
Reshapes Treatment Decisions
Pre-NAPOLI-3 Era:
Uncertainty
Treatment
selection was based on:
– Indirect
cross-trial comparisons
– FOLFIRINOX
assumed superior to nab-paclitaxel/gemcitabine (unproven)
–
Nab-paclitaxel/gemcitabine preferred for older/frailer patients
–
Equipoise-driven rather than evidence-driven
Post-NAPOLI-3
Era: Evidence-Based Hierarchy
Now
clinicians have definitive comparative data establishing clear treatment
ranking.
NALIRIFOX vs
FOLFIRINOX: Indirect Comparison
While
no direct head-to-head trial exists, indirect comparison using PRODIGE 4 data
reveals:
|
Parameter |
NALIRIFOX |
FOLFIRINOX |
Advantage |
|
Median OS |
11.1 mo |
11.1 mo |
Equivalent |
|
Median PFS |
7.4 mo |
6.4 mo |
NALIRIFOX (+1 mo) ⭐ |
|
ORR |
41.8% |
31.6% |
NALIRIFOX (+10%) ⭐ |
|
Grade 3-4 Neutropenia |
14% |
45.7% |
NALIRIFOX (68% reduction) ⭐⭐ |
|
Grade 3-4 Neuropathy |
3% |
9% |
NALIRIFOX (67% reduction) ⭐⭐ |
Clinical Translation: – Equivalent
survival but better disease control (PFS/ORR) – Markedly superior
tolerability (3-fold lower hematological toxicity)
–
NALIRIFOX emerges as potentially preferable to FOLFIRINOX pending direct
comparative trials
Source: Wainberg ZA, et al. JAMA Netw Open. 2023;6(12):e2349321
Evidence-Based
Treatment Hierarchy for Metastatic PDAC
TIER 1: Preferred for
ECOG 0-1
✅ NALIRIFOX
– Superior OS/PFS vs nab-paclitaxel/gemcitabine; EMA-approved; manageable
toxicity
✅ FOLFIRINOX
– Comparable OS; established option; longer track record; greater toxicity
TIER 2:
Alternative for ECOG 0-1 or Selected ECOG 2
⚠️
Nab-paclitaxel/Gemcitabine – Proven efficacy; appropriate for patients
declining quadruplet intensity or with contraindications
TIER 3: Palliative Intent
⚖️ Gemcitabine
monotherapy – ECOG 2-3 or severe comorbidities
🇬🇧 UK NHS
Practice Implementation
Commissioning Pathway
NALIRIFOX entered NHS England
commissioning evaluation following EMA approval (early 2024):
Current Status: –
✅ EMA approval granted
⏳ NICE Health Technology Assessment
(HTA) underway
⏳ Cancer Drugs Fund (CDF) pathway under
consideration
📊
Real-world effectiveness data collection planned
Implementation Requirements
1. Patient Selection Criteria
– ECOG performance status 0-1
– Histologically/cytologically
confirmed metastatic PDAC
– Adequate renal function (eGFR
>40 mL/min if possible)
– Adequate hepatic function
(bilirubin <2x ULN)
– No peripheral neuropathy grade
≥2
2. Baseline Assessments
✓ Baseline CBC, comprehensive
metabolic panel
✓ Cardiac assessment (LVEF if
indicated)
✓ Tumor imaging (CT
chest/abdomen/pelvis ± baseline CA 19-9)
✓ Molecular profiling (BRCA, MMR
status)
3. Supportive Care
Infrastructure
– Anti-diarrheal prophylaxis
protocols
– Weekly electrolyte monitoring
(first 8 weeks)
– Anti-emetic regimens optimized
– Hydration support access
– Rapid access to oncology triage
for grade ≥3 events
4. Multidisciplinary Team
(MDT) Discussion
All metastatic PDAC patients
should be discussed at hepatopancreatobiliary MDT to determine NALIRIFOX
suitability, considering:
– Comorbidities (cardiac, renal,
hepatic)
– Patient functional status
– Patient treatment preferences
– Contraindications
(absolute/relative)
5. Shared Decision-Making
Conversations Counsel patients on treatment options
with transparent discussion:
|
Factor |
NALIRIFOX |
FOLFIRINOX |
Nab-pac/Gem |
|
Survival |
Best evidence |
Equivalent |
Shorter |
|
Disease Control (PFS) |
7.4 mo |
6.4 mo |
5.6 mo |
|
Response Rate |
41.8% |
31.6% |
36.2% |
|
Diarrhea Risk |
Moderate |
Low |
Low |
|
Neuropathy Risk |
Low |
High |
Moderate |
|
Infusion Duration |
46 hours/cycle |
46 hours/cycle |
30 min/cycle |
|
Clinic Visits |
Every 2 weeks |
Every 2 weeks |
3x weekly |
Quality of Life
Considerations
Until formal NAPOLI-3 quality
of life analysis publication, clinical inference suggests NALIRIFOX may
preserve QoL through:
•
Reduced neurotoxicity → Preserved manual dexterity, mobility, independence
•
Longer tolerability → Extended treatment duration enabling sustained therapy
•
Lower infection risk → Fewer hospitalizations from febrile neutropenia
•
Trade-off: Increased diarrhea requiring proactive management and patient
education
UK Nursing Role: Oncology nurses provide critical diarrhea self-management
education, hydration counseling, and early warning symptom monitoring to
prevent avoidable hospital admissions.
Equity & Access
Considerations
Representation in NAPOLI-3:
⚠️ 82-84% White (limited ethnic
diversity)
⚠️ Predominantly North
American/European
✅ No upper age limit (supports
treating older adults with good PS)
Recommendations:
1. Ensure equitable NALIRIFOX
access regardless of ethnicity
2. Prioritize inclusion of BAME
communities in future pancreatic cancer research
3. Challenge ageist
assumptions—chronological age ≠ treatment ineligibility
4. Monitor real-world outcomes
across diverse populations post-approval
🔬 Study Strengths
& Limitations
Methodological Strengths
✅ First direct head-to-head
comparison in first-line metastatic PDAC
✅ Global, multicentre design
(187 sites, 18 countries)
✅ Pragmatic eligibility (no
upper age limit; real-world populations)
✅ Robust statistical framework
(90% power; hierarchical testing)
✅ Comprehensive safety data
(standardized CTCAE 5.0 coding)
✅ Independent oversight (Data
Monitoring Committee)
Methodological Limitations
⚠️ Open-label design –
Lacks blinding; potential bias in symptom attribution/supportive care intensity
(mitigated by blinded RECIST assessment)
⚠️ No quality of life primary
endpoint – PRO collection exploratory; formal QoL analysis pending
⚠️ Limited biomarker
characterization – Tissue/serum collected but biomarker-driven subgroup
analyses not pre-specified
⚠️ Predominantly Caucasian
(82-84%) – Limited generalizability to ethnic minorities with distinct
pancreatic cancer biology
⚠️ Excludes ECOG 2-4 patients
– Frail populations underrepresented
❓ Unanswered Clinical
Questions
1.
Optimal treatment duration: When to stop NALIRIFOX? Continuous vs time-limited strategies?
2.
Maintenance therapy
approach: Does de-escalation to fluoropyrimidine
maintenance after 4-6 months preserve efficacy while reducing toxicity?
3.
Biomarker predictive
utility: Which molecular features predict NALIRIFOX
benefit? (BRCA mutations, HRD, MSI-H)
4.
Direct NALIRIFOX vs
FOLFIRINOX comparison: Phase 3 head-to-head trial
needed to establish optimal quadruplet regimen
5.
Second-line therapy after
NALIRIFOX: Optimal salvage regimens after NALIRIFOX
progression?
6.
Quality of life trade-offs: Formal PRO analysis comparing patient-reported tolerability and QoL
impacts?
7.
Economic sustainability: Cost-effectiveness from UK NHS perspective (pending NICE HTA)?
📚 Key Evidence Comparison
NAPOLI-3 vs
Major First-Line Pancreatic Cancer Trials
|
Trial |
Year |
Regimen |
N |
Median OS |
Comparator OS |
HR |
Trial Design |
|
PRODIGE 4/ACCORD 11 |
2011 |
FOLFIRINOX |
342 |
11.1 mo |
Gem 6.8 mo |
0.57 |
vs Monotherapy |
|
MPACT |
2013 |
Nab-pac/Gem |
861 |
8.5 mo |
Gem 6.7 mo |
0.72 |
vs Monotherapy |
|
NAPOLI-3 |
2023 |
NALIRIFOX |
770 |
11.1 mo |
Nab-pac/Gem 9.2 mo |
0.83 |
Head-to-head ⭐ |
Landmark Achievement: NAPOLI-3 is the first
phase 3 trial directly comparing two active combination regimens—filling an
12-year evidence gap.
✅ Summary
Recommendations for UK NHS Clinical Practice if NICE approved
DO:
✅ Offer NALIRIFOX as preferred
first-line therapy for ECOG 0-1 metastatic PDAC patients (level 1 evidence)
✅ Maintain FOLFIRINOX as alternative
option for patients unable to access NALIRIFOX (comparable efficacy; longer
track record)
✅ Implement proactive supportive
care (anti-diarrheal prophylaxis, electrolyte monitoring, anti-emetics)
✅ Conduct upfront molecular
profiling (BRCA, MMR, KRAS G12C status)
✅ Engage patients in shared
decision-making with transparent efficacy/toxicity discussion
✅ Ensure equitable access
across demographic groups and age ranges
DON’T:
❌ Automatically reserve quadruplet
therapy for “fit” patients—extended median age (65 years) supports broad
applicability
❌ Assume nab-paclitaxel/gemcitabine
is equivalent to NALIRIFOX (NAPOLI-3 establishes superiority)
❌ Overlook peripheral neuropathy
reduction—a quality-of-life advantage often underappreciated
❌ Exclude older adults based on
chronological age alone (ECOG PS is appropriate selector)
🎯 Clinical Bottom Line
NAPOLI-3
establishes NALIRIFOX as a new evidence-based first-line standard of care for
metastatic pancreatic cancer, providing:
•
Superior overall survival (11.1 vs 9.2 months; 18-month OS 26.2% vs 19.3%)
•
Doubled 12-month PFS (27.4% vs 13.9%)
•
Paradoxically superior
safety (lower hematological toxicity despite
quadruplet composition)
•
FDA/EMA regulatory approval (early 2024)
📖 Key References
Wainberg ZA, Melisi D,
Macarulla T, et al. NALIRIFOX versus nab-paclitaxel and gemcitabine in
treatment-naive patients with metastatic pancreatic ductal adenocarcinoma
(NAPOLI 3): a randomised, open-label, phase 3 trial. Lancet.
2023;402(10409):1272-1281. https://doi.org/10.1016/S0140-6736(23)01366-1
Plain language summary of
NALIRIFOX compared with nab-paclitaxel and gemcitabine for patients with
metastatic pancreatic cancer (NAPOLI 3). Future Oncol. 2025.
https://doi.org/10.1080/14796694.2025.2458458
Systemic Therapy for
Metastatic Pancreatic Cancer—Current Landscape and Future Directions. Cancers.
2024;16:3657. https://doi.org/10.3390/cancers16103657
Management of Metastatic
Pancreatic Cancer—Comparison of Global Guidelines over the Last 5 Years. Cancers.
2023;15(17):4400. https://doi.org/10.3390/cancers15174400
Prognosis and Treatment of
Gastric Cancer: A 2024 Update. Cancers. 2024;16(9):1708.
https://doi.org/10.3390/cancers16091708
Consensus, debate, and
prospective on pancreatic cancer treatments. Oncology. 2024.
NALIRIFOX, FOLFIRINOX, and
Gemcitabine With Nab-Paclitaxel as First-Line Chemotherapy for Metastatic
Pancreatic Cancer. JAMA Netw Open. 2023;6(12):e2349321.
https://doi.org/10.1001/jamanetworkopen.2023.49321