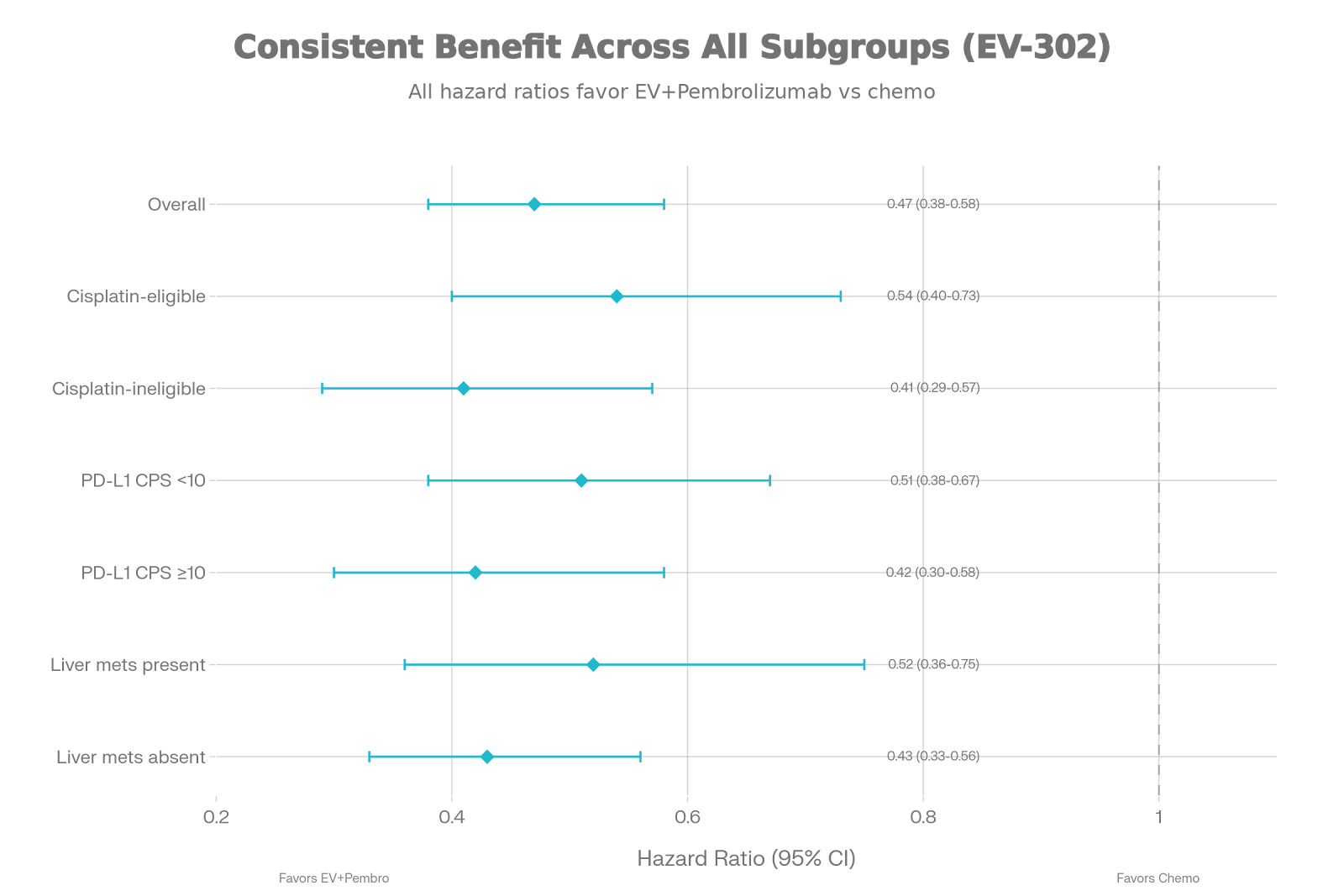
Predicting Treatment Success with Lutetium-177-PSMA: New Nomograms from the VISION Trial
Treatment
selection in advanced prostate cancer just got sharper.
Lutetium-177-PSMA-617 (Pluvicto®)
has changed the landscape for metastatic castration-resistant prostate cancer
(mCRPC). But with high costs, potential toxicity, and variable responses, the
million-dollar question remains: Who
will actually benefit?
New data published in eClinicalMedicine
(October 2024) gives us the first high-quality, validated nomograms to answer
this question. Built from the landmark Phase 3 VISION trial, these tools allow
us to predict individual patient outcomes with far greater accuracy than ever
before.
The Bottom Line (5 Key Takeaways)
·
First Prospective Tools: These are the first predictive models
built from large-scale, prospective Phase 3 data (VISION trial), making them
the new “gold standard” over previous retrospective models.
·
High Accuracy: The nomograms achieved impressive predictive accuracy
(C-index 0.73 for Overall Survival),
significantly outperforming older tools.
·
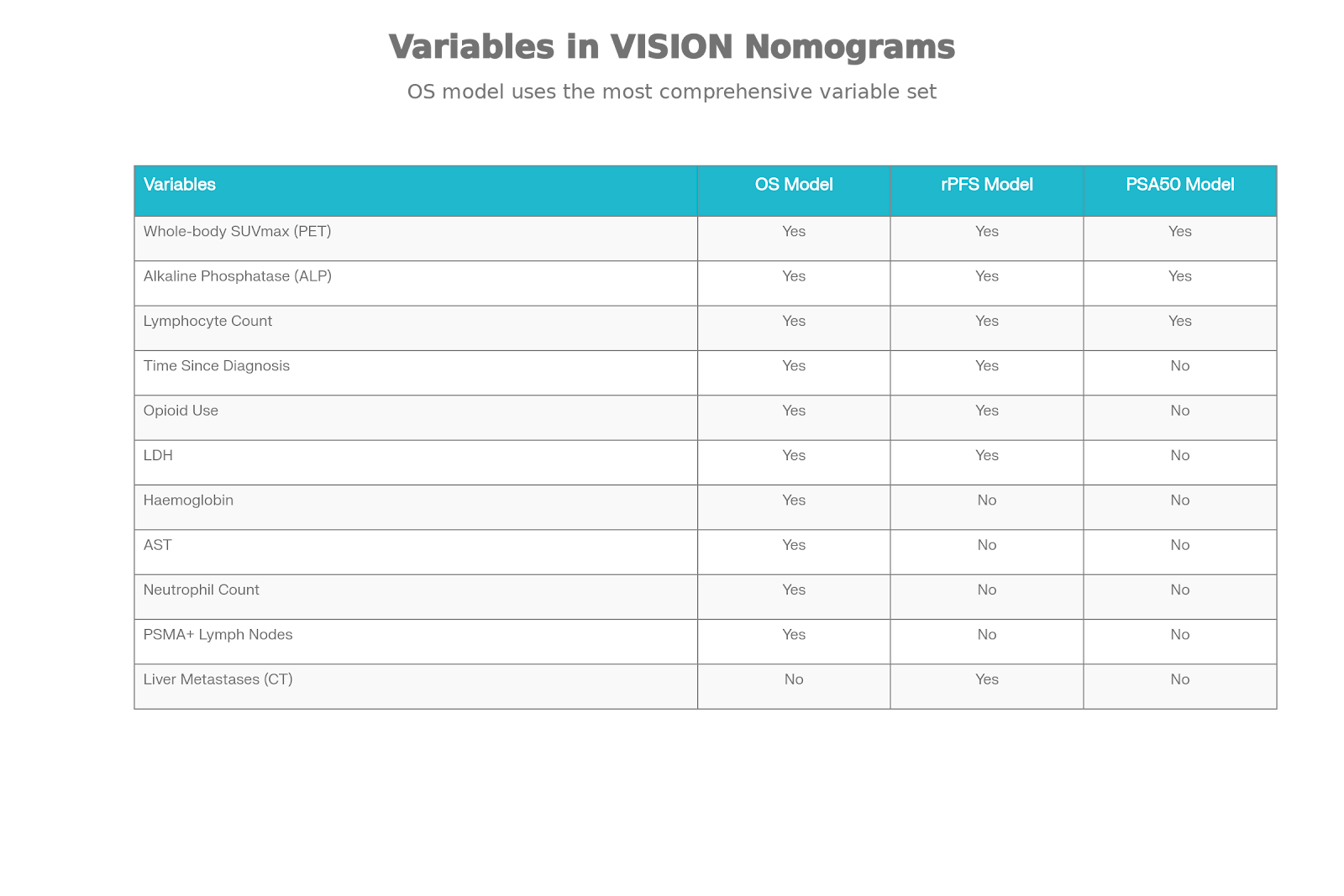
More Than Just PET: Success isn’t just about SUVmax.
Clinical factors like hemoglobin, LDH,
and time since diagnosis are equally critical drivers of survival.
·
Accessible for All: You don’t need advanced PET
quantification software. The survival models remain highly accurate using
standard clinical variables, making them usable in any NHS center.
·
Actionable: These tools can help you enrich clinical trials, counsel
patients on realistic expectations, and avoid futile toxicity in those unlikely
to respond.
·
Source: Analysis of the VISION
Phase 3 Trial (n=551 patients treated with 177Lu-PSMA-617).
·
Patient Profile: Heavily pre-treated mCRPC (post-ARPI
& post-taxane), PSMA-positive.
·
Goal: Create mathematical models (nomograms) to predict three key
outcomes:
a. Overall
Survival (OS)
b. Radiographic
Progression-Free Survival (rPFS)
c. PSA50
Response (≥50% decline)
Key Findings: What Predicts Success?
The researchers developed three complementary nomograms. The
performance was impressive, validating that we can reliably identify
“super-responders” versus those with poor prognosis.
1. Overall Survival (OS) Nomogram
·
Accuracy: C-index 0.73
(Strong prediction)
·
What Drives It? A mix of tumor biology and host
reserve.
o Good
signs: High SUVmax, long time since
diagnosis.
o Bad
signs: Opioid use, high LDH/ALP/AST,
low hemoglobin/lymphocytes, lymph node burden.
2. Radiographic PFS (rPFS) Nomogram
·
Accuracy: C-index 0.68
·
What Drives It? Similar to OS but heavily influenced
by liver metastases and liver
enzymes (LDH, ALP), reflecting disease burden.
·
Accuracy: AUC 0.72
·
What Drives It? Surprisingly simple. Short-term PSA
drop is almost entirely driven by PSMA
expression (SUVmax) and disease burden (ALP, lymphocytes).
Variables Driving Prediction in VISION Nomograms
When tested head-to-head against older models (like the 2021
Gafita nomograms), the VISION models won easily. This proves that building
tools from standardized, high-quality clinical trial data yields far better
real-world reliability.
Clinical Application: How to Use This
Tomorrow
Use the nomograms to give patients personalized estimates.
·
Scenario A: A patient with high SUVmax, good marrow reserve, and no
opioids might have a predicted 24-month survival of 85%.
·
Scenario B: A patient with liver mets, low hemoglobin, and high LDH
might have a predicted 24-month survival of only 30%, shifting the conversation toward palliative care or clinical
trials.
If you are deciding between Cabazitaxel and
Lutetium-177-PSMA:
·
Favorable Nomogram Score → Prioritize Lutetium (high chance of
response).
·
Unfavorable Score → Consider Cabazitaxel or other
alternatives.
3. “No PET Software? No Problem.”
Crucially for many NHS centers: The survival prediction models work just as well without SUVmax data.
If you don’t have advanced PET quantification software, you can still use the
clinical variables (Labs + History) to get an accurate prognosis.
Implications for UK NHS Practice
With Lutetium-177-PSMA-617 currently navigating NICE
appraisal and available via specific access schemes (trials, compassionate use,
IFRs), these tools are vital:
·
Support Funding Requests (IFRs): Use the nomogram score to objectively
demonstrate that your patient is a “predicted responder,”
strengthening your case for funding.
·
Trial Recruitment: Use the tools to identify ideal
candidates for ongoing UK trials (e.g., combinations with immunotherapy).
·
Resource Management: In a cost-constrained system, avoiding
expensive therapy in patients destined not to benefit is just as important as
treating those who will.
·
Post-Hoc: These are retrospective analyses of a prospective trial;
they need prospective validation in a real-world NHS cohort.
·
PSMA+ Only: The VISION trial excluded patients with PSMA-negative
lesions. These tools cannot be used
for unselected patients (you still need a PET scan to confirm eligibility
first).
·
Late Stage: Validated only for post-taxane, post-ARPI patients. Do not
use for hormone-sensitive disease.
of outcomes with [177Lu]Lu-PSMA-617: analysis of the phase 3 VISION trial. eClinicalMedicine. 2024 Oct 4. DOI:
10.1016/j.eclinm.2024.102862