2025 updates to TOPAZ-1
·
Title: Durvalumab plus chemotherapy in advanced biliary tract
cancer: 3-year overall survival update from the phase III TOPAZ-1 study
·
Authors: Do-Youn Oh, A.R. He, et al.
·
Journal: Journal of Hepatology
·
Publication Date: April 2025 (ePub)
·
DOI: 10.1016/j.jhep.2025.02.019 (approximate based on recent
listing)
·
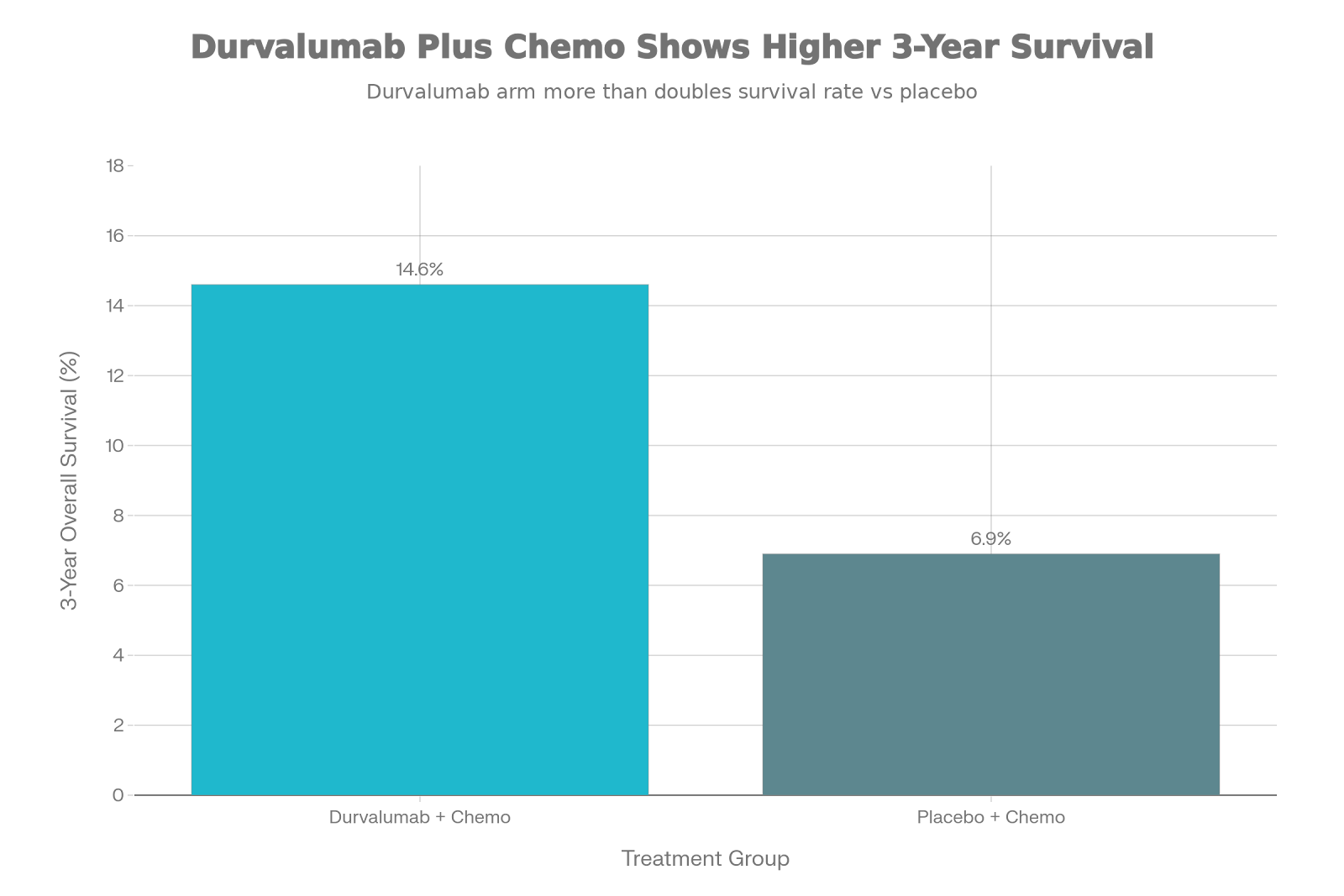
Doubled Survival Rate: Durvalumab plus chemotherapy more than doubled the 3-year
overall survival rate (14.6% vs. 6.9%) compared to chemotherapy alone.
·
Sustained Benefit: The survival curves remain separated at 3 years, confirming
the durability of the immunotherapy response.
·
Safe Long-term: No new or cumulative toxicity signals emerged after 3 years
of follow-up.
·
Universal Efficacy: The benefit was consistent across tumor locations
(intrahepatic, extrahepatic, gallbladder) and PD-L1 status.
·
New Baseline: This data establishes the 14-15% 3-year survival mark as
the new benchmark for future first-line BTC trials.
For decades, treating advanced biliary
tract cancer (BTC) felt like hitting a ceiling. We relied on the ABC-02 regimen
(gemcitabine and cisplatin), and while it worked, the clock was always ticking
loud and fast. Most patients faced a median survival of less than a year.
The TOPAZ-1 trial changed the conversation in 2022, but the skeptics
asked: “Is the benefit real? Does it
last?”
The 2025 update published in the Journal
of Hepatology gives us a resounding YES.
The Headline: Breaking the 10% Barrier
The most striking finding from this
year’s update is the “tail” of
the survival curve. In the world of immuno-oncology, we look for that
plateau—the group of patients who don’t just survive a few extra months, but
years.
Look at the difference at the 3-year
mark:
·
Chemotherapy alone: Only 6.9% of patients were alive.
·
Durvalumab + Chemo: 14.6%
of patients were alive.
That is more than double. For a cancer
this aggressive, reaching a point where nearly 1 in 7 patients is alive at 3 years is a massive leap forward.
Why This Matters for Your Monday Clinic
When you sit down with a newly
diagnosed patient this week, your counseling can shift.
·
Before: We offered chemotherapy to “slow things down.”
·
Now: We offer chemo-immunotherapy with the genuine hope that
they might be in that ~15% of “super-responders” who achieve
long-term control.
The “Super-Responder” Phenotype
Interestingly, this benefit wasn’t
restricted to just one type of anatomy. Whether your patient has a gallbladder
tumor or an intrahepatic cholangiocarcinoma, the benefit stands. We still don’t
have a perfect biomarker (PD-L1 levels didn’t perfectly predict response), so
for now, all fit comers should be
offered this standard of care.
The 3-year data also reassured us that
adding immunotherapy doesn’t add a heavy burden of late toxicity. If your
patient tolerates the first few cycles well, they are unlikely to run into new
surprises years down the line.
The
Bottom Line: The
TOPAZ-1 regimen isn’t just a statistical win; it’s a clinical win. It has
raised the bar and finally given us a “long tail” of survival to aim
for.
For more
oncology updates and deep dives, keep checking dpsoncology.blogspot.com.