Claudin 18.2-Targeted Drugs: Regulatory Status and Clinical Development
FDA and EMA Approved Drugs
Zolbetuximab
(Vyloy®) – Astellas Pharma
The ONLY Claudin 18.2-targeted therapy currently approved by FDA and
EMA
Regulatory Approvals:
•
Japan (MHLW): March 2024 ✅ – First approval worldwide
•
UK (MHRA): August 14, 2024 ✅
•
European Union (EMA): September 20, 2024 ✅
•
South Korea (MFDS): September 2024 ✅
•
United States (FDA): October 18, 2024 ✅
•
China (NMPA): January 2025 ✅
NICE (UK) Status:
❌ NOT RECOMMENDED –
March 12, 2025 (TA1046)
Reason for rejection:
•
Cost-effectiveness concerns –
ICER (incremental cost-effectiveness ratio) exceeded £20,000 per QALY threshold
•
NICE concluded that
zolbetuximab plus chemotherapy is less costly but also less effective
compared to nivolumab or pembrolizumab plus chemotherapy
•
Economic modeling did not
justify the cost relative to existing alternatives
•
Despite clinical benefit in
trials, the committee determined it does not represent acceptable use of NHS
resources
Implication: Zolbetuximab is licensed in the UK but will NOT be routinely
funded by NHS England, Wales, Scotland, or Northern Ireland
Approved Indication:
First-line treatment
of adults with:
•
Locally advanced
unresectable OR metastatic
•
HER2-negative
•
CLDN18.2-positive (defined as ≥75% of tumor cells with moderate-to-strong membranous
staining)
•
Gastric or gastroesophageal
junction (GEJ) adenocarcinoma
Given in
combination with: Fluoropyrimidine- and
platinum-containing chemotherapy (mFOLFOX6 or CAPOX)
Supporting Clinical Trials:
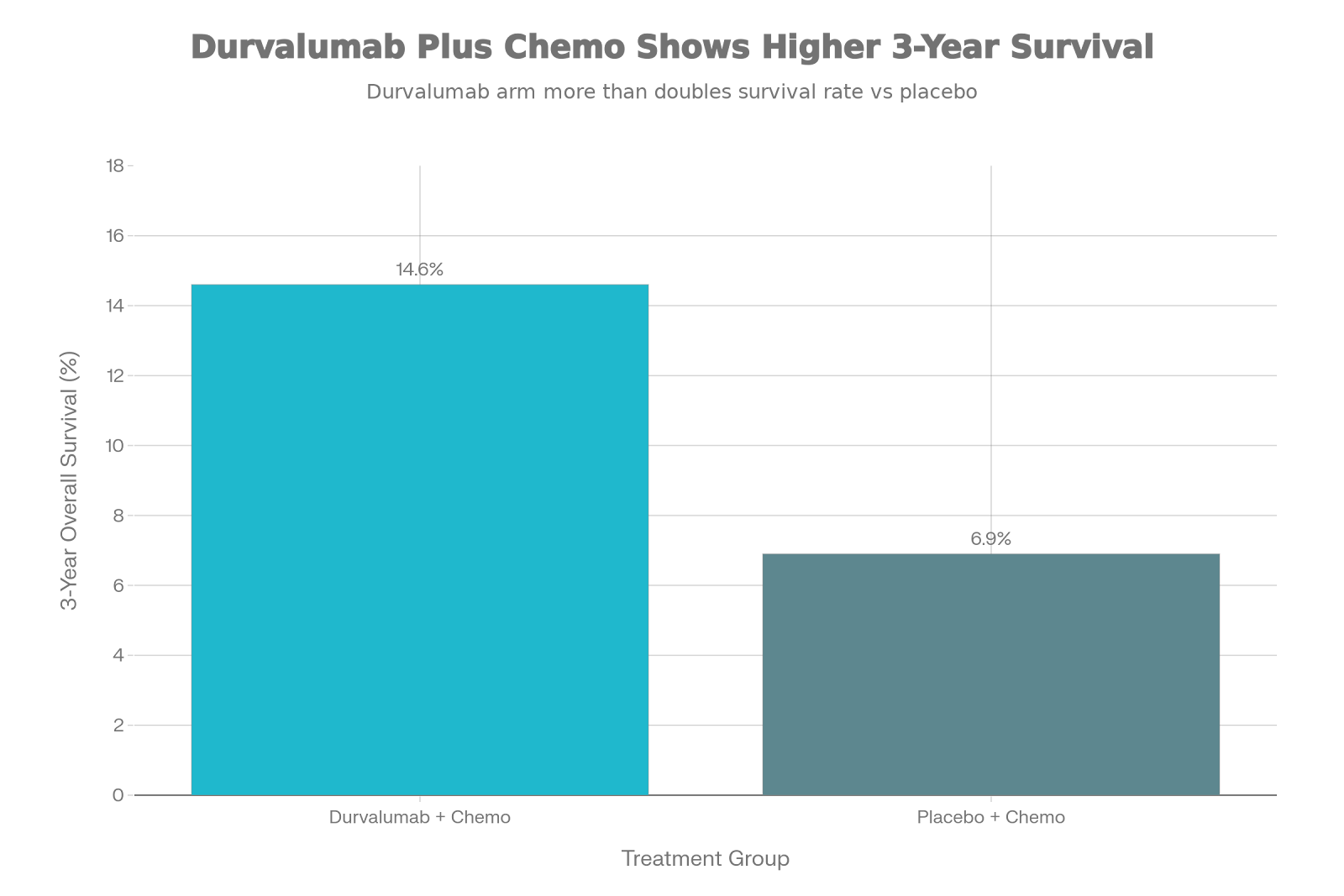
1.
SPOTLIGHT Trial
(NCT03504397): Zolbetuximab + mFOLFOX6 vs. placebo
+ mFOLFOX6
–
Median OS: 14.39 months vs.
12.16 months (HR 0.77; P=0.0053)
–
Median PFS: 10.61 months vs.
8.67 months (HR 0.75; P=0.0066)
–
24-month OS: 38.8% vs. 28.4%
2.
GLOW Trial (NCT03462719): Zolbetuximab + CAPOX vs. placebo + CAPOX
–
Median OS: 14.39 months vs.
12.16 months (HR 0.77; P=0.0053)
–
Median PFS: 8.21 months vs.
6.80 months
–
Consistent benefit across
subgroups
Companion Diagnostic:
VENTANA CLDN18
(43-14A) RxDx Assay (Roche)
•
CE Mark approved October 9,
2024
•
FDA-approved companion
diagnostic
•
Identifies patients eligible
for zolbetuximab treatment
•
~38% of screened gastric/GEJ
cancer patients test CLDN18.2-positive
Dosing:
•
Loading doses: 800 mg IV on Cycle 1 Day 1; 600 mg IV on Cycle 1 Days 8 and 15
•
Maintenance: 600 mg IV every 3 weeks (with mFOLFOX6) OR 800 mg IV every 2 weeks
(with CAPOX)
•
Continue until disease
progression or unacceptable toxicity
Most Common Adverse Events:
•
Nausea (82-88%)
•
Vomiting (67-78%)
•
Decreased appetite (44-49%)
•
Most AEs are grade 1-2 and
manageable with antiemetics and supportive care
Drugs in
Late-Stage Clinical Development
1. CT041
(Satri-cel / Satricabtagene Autoleucel) – CARsgen Therapeutics
Type: CLDN18.2-targeted autologous CAR-T
cell therapy
Regulatory Designations:
•
China NMPA: Breakthrough Therapy Designation (March 2025) + Priority Review
(May 2025)
•
US FDA: Regenerative Medicine Advanced Therapy (RMAT) designation (January
2022)
•
US FDA: Orphan Drug Designation (September 2020)
•
EMA: PRIME designation
Development Status:
•
NDA submission to China NMPA: Expected June 2025
•
Potential approval: Could become world’s first approved CAR-T for solid tumors in 2025
•
Phase II pivotal trial
published: The Lancet (June 2025)
Indication:
Treatment of CLDN18.2-positive
advanced gastric/gastroesophageal junction adenocarcinoma in patients who have
failed ≥2 prior lines of therapy
Key Efficacy Data
(Phase II CT041-ST-01):
•
Objective Response Rate
(ORR): 54.9%
•
Disease Control Rate (DCR): 96.1%
•
Median Overall Survival: 9.0 months (high CLDN18.2 expression subgroup: 13.1 months)
•
Median PFS: 6.9 months
•
Superior to chemotherapy
comparator arm
Safety:
•
Manageable toxicity profile
•
Cytokine release syndrome (CRS)
observed but manageable
•
On-target off-tumor gastric
toxicity remains a challenge being addressed through AND-gate CAR technology
2. IBI343 –
Innovent Biologics / Takeda (October 2025 partnership)
Type: CLDN18.2-targeted antibody-drug
conjugate (ADC) – Anti-CLDN18.2 mAb conjugated to exatecan (TOPO1 inhibitor)
Regulatory Designations:
•
China NMPA: Breakthrough Therapy Designation (May 2024) for gastric cancer
•
US FDA: Fast Track Designation (FTD) for advanced pancreatic cancer
•
US FDA: IND approved for PDAC
Development Status:
•
Phase 3 trial: Ongoing in previously treated gastric cancer (Japan and China) –
NCT06238843 (G-HOPE-001)
•
Phase 3 trial: In preparation for first-line gastric cancer
•
Phase 1 results: Presented at ASCO 2024
Indications Being Studied:
1.
Gastric or GEJ adenocarcinoma
(CLDN18.2+) – post 2 prior lines of therapy
2.
Advanced pancreatic ductal
adenocarcinoma (PDAC) – relapsed/refractory after 1 prior line
Key Efficacy
Data (Phase 1 – Pancreatic Cancer):
•
ORR: 40% (in 6 mg/kg dose group, CLDN18.2 ≥60% patients)
•
Encouraging efficacy in
difficult-to-treat late-line pancreatic cancer
Key Efficacy Data
(Gastric Cancer):
•
Over 340 patients treated with
IBI343 across trials
•
Tolerable safety and promising
efficacy signals
3. AZD0901 –
AstraZeneca
Type: CLDN18.2-targeted antibody-drug
conjugate (ADC) – Anti-CLDN18.2 mAb conjugated to monomethyl auristatin E
(MMAE)
Development Status:
•
Phase 3 CLARITY-Gastric 01
trial: Enrolling (NCT06346392) – launched 2024
•
Phase 2 CLARITY-PanTumour01
trial: Ongoing (NCT06219941)
Phase 3 Trial
Design (CLARITY-Gastric 01):
•
Population: Advanced unresectable/metastatic GC/GEJC (non-HER2+, CLDN18.2+)
•
Setting: Second-line or later (≥1 prior platinum-fluoropyrimidine regimen)
•
Design: Randomized 1:1:1 to AZD0901 dose level 1 vs. dose level 2 vs.
investigator’s choice (ramucirumab + paclitaxel, paclitaxel, docetaxel,
irinotecan, TAS-102, apatinib)
•
Primary endpoints: PFS (ITT population) and OS (3L+ population)
•
Geographic scope: 16 countries across Asia, Europe, North America
Phase 1 Data
(Published J Clin Oncol 2023):
•
Demonstrated clinical activity
in advanced GC/GEJC
•
Dose escalation/expansion
ongoing
Indications Being Studied:
1.
Gastric/GEJ cancer (2L+
setting)
2.
Pancreatic cancer (combination
with chemotherapy)
3.
Biliary tract cancer
(monotherapy)
4. JS107 –
Shanghai Junshi Biosciences
Type: CLDN18.2-targeted antibody-drug
conjugate (ADC) – Humanized anti-CLDN18.2 mAb conjugated to monomethyl
auristatin E (MMAE)
Development Status:
•
Phase III trial: Planned for 2026
•
Phase I data: Presented at ESMO Asia Congress 2025 (December)
Key Efficacy
Data (Phase I – First-line G/GEJA):
Combination: JS107 + toripalimab (anti-PD-1) + XELOX chemotherapy
•
ORR: 86.7% in high CLDN18.2 expression patients (≥40% cells with ≥2+
staining)
•
Disease Control Rate: 100%
•
Median PFS: 11.14 months
•
Safety: Grade ≥3 TRAEs in 55.6–58.3%, primarily hematologic
Unique Features:
•
Shows activity even in low
CLDN18.2 expression tumors (5% TC with <2+ staining achieved 1 CR + 1 PR
in phase I)
•
Triple mechanism: ADCC, CDC,
and MMAE-mediated cytotoxicity with bystander killing effect
5. AMG 910
(Gresonitamab) – Amgen
Type: Bispecific T-cell engager (BiTE®)
– Anti-CLDN18.2 / Anti-CD3
Development Status:
•
Phase 1 trial: Ongoing (NCT04260191)
•
Half-life extended (HLE) BiTE
molecule
Mechanism:
Redirects cytotoxic CD8+ and
CD4+ T cells to kill CLDN18.2-expressing cells
Indication Being Studied:
Metastatic or
locally advanced unresectable gastric/GEJ adenocarcinoma (CLDN18.2+) –
refractory/relapsed after ≥2 prior lines
Trial Design:
•
Dose escalation/expansion study
•
Evaluating safety, MTD/RP2D,
PK, PD, and preliminary efficacy
Other Claudin
18.2 Drugs in Clinical Pipeline
ADCs (Antibody-Drug
Conjugates):
1.
CMG901 – Keymed Biosciences / Lepu Biopharma (Phase I – NCT04805307)
–
Anti-CLDN18.2 mAb + MMAE
–
First CLDN18.2 ADC globally
2.
BMS-986476 – Bristol Myers Squibb (Phase I/II)
3.
TPX4589 – Turning Point Therapeutics (Preclinical/Phase I)
4.
ASKB589 – Ascletis Pharma (Phase I)
5.
SKB315 – Jiangsu Simcere Pharmaceutical (Phase I)
6.
Arcotatug tavatecan – Phase III
7.
Garetatug rezetecan – Phase III
8.
Sonesitatug vedotin – Phase III
9.
Tecotabart vedotin – Phase III
Bispecific Antibodies:
1.
AZD5863 – AstraZeneca (CLDN18.2/CD3) – Phase I (NCT06005493)
2.
ABL111 – ABL Bio (CLDN18.2/4-1BB + PD-1 inhibitor) – Phase I
3.
ATG022/ATN022 – Antengene (CLDN18.2/CD3) – Phase I in China (approved March 2023)
4.
QLS31905 – Chinese developer (CLDN18.2/CD3 bispecific)
5.
PT886 (Spevatamig) – Phase I
CAR-T Therapies:
1.
CT041 (Satri-cel) – CARsgen [see detailed section above]
2.
VHH-based CAR-T – Humanized anti-CLDN18.2 VHH CAR-T (preclinical/early phase)
3.
Dual-targeted CAR-T – FAP + CLDN18.2 (preclinical)
4.
AND-gate CAR-T – CLDN18.2 + Mesothelin (preclinical – addressing on-target
off-tumor toxicity)
Monoclonal Antibodies:
1.
Osemitamab – Phase II (TranStar 301 Phase III planned with chemotherapy +
nivolumab)
2.
ZL-1211 – Targets GC cells across both high and low CLDN18.2 expression
Key Clinical Considerations
CLDN18.2 Expression Cutoffs:
Different drugs use different
definitions:
•
Zolbetuximab: ≥75% of tumor cells with moderate-to-strong (≥2+) membranous
staining
•
Most ADCs and CAR-T: Variable cutoffs (40-75% TC with ≥1+ or ≥2+ staining)
•
Prevalence: ~38% of gastric/GEJ cancers meet ≥75% cutoff; up to 50-70% have
some CLDN18.2 expression
CLDN18.2 Expression by
Cancer Type:
•
Gastric/GEJ adenocarcinoma: 40-80%
•
Pancreatic ductal
adenocarcinoma: 50-70%
•
Esophageal adenocarcinoma: 30-50%
•
Intrahepatic
cholangiocarcinoma: 20-30%
•
Colorectal cancer: Lower expression (~10-20%)
Testing Requirements:
•
Immunohistochemistry (IHC)
companion diagnostic required
•
VENTANA CLDN18 (43-14A) RxDx
Assay is FDA/CE Mark approved for zolbetuximab
•
Testing should become routine
in all gastric/GEJ adenocarcinomas
Summary Table:
Claudin 18.2 Drugs Regulatory Status
|
Drug |
Type |
Company |
FDA Status |
EMA Status |
NICE Status |
Expected Approval |
|
Zolbetuximab (Vyloy) |
mAb |
Astellas |
✅ Approved Oct 2024 |
✅ Approved Sep 2024 |
❌ Not Recommended (Mar 2025) |
Already approved |
|
CT041 (Satri-cel) |
CAR-T |
CARsgen |
RMAT designation |
PRIME |
Not yet assessed |
China: Mid-2025; US/EU: 2026-2027 |
|
IBI343 |
ADC |
Innovent/Takeda |
Fast Track (PDAC) |
Not yet submitted |
Not yet assessed |
2026-2027 |
|
AZD0901 |
ADC |
AstraZeneca |
Phase 3 ongoing |
Phase 3 ongoing |
Not yet assessed |
2027-2028 |
|
JS107 |
ADC |
Junshi |
Phase 3 planning |
Phase 3 planning |
Not yet assessed |
2027-2028 |
|
AMG 910 |
BiTE |
Amgen |
Phase 1 |
Phase 1 |
Not yet assessed |
2028+ |
Key Takeaways for
Clinical Practice
1.
Zolbetuximab is the only
approved CLDN18.2 drug but faces reimbursement
challenges in the UK due to cost-effectiveness concerns
2.
CLDN18.2 testing should be
routine for all gastric/GEJ adenocarcinomas at
diagnosis
3.
Multiple drug classes are in
development: monoclonal antibodies, ADCs,
bispecific antibodies, and CAR-T therapies
4.
CT041 CAR-T could become the first approved CAR-T for solid tumors globally
(China approval expected mid-2025)
5.
ADC landscape is crowded: At least 10 CLDN18.2 ADCs in clinical development, with IBI343,
AZD0901, and JS107 leading in phase III trials
6.
Efficacy signals are
promising across all modalities, but safety
management (especially GI toxicity) remains critical
7.
Combination strategies (CLDN18.2-targeted therapy + immunotherapy + chemotherapy) show the
highest response rates (80-87% ORR)
8.
Watch for:
–
CT041 approval in China (June
2025)
–
Phase 3 readouts for IBI343,
AZD0901, and JS107 (2026-2027)
–
NICE reconsideration of
zolbetuximab if pricing agreements change
Date
compiled: January 5, 2026
Sources: FDA, EMA, NICE, MHRA, NMPA press releases; ClinicalTrials.gov;
ESMO 2025 Congress abstracts; published literature