ESMO Oncology Weekly Digest — 8–15 February 2026
ESMO
Oncology Weekly Digest — 8–15 February 2026
Key Takeaways
•
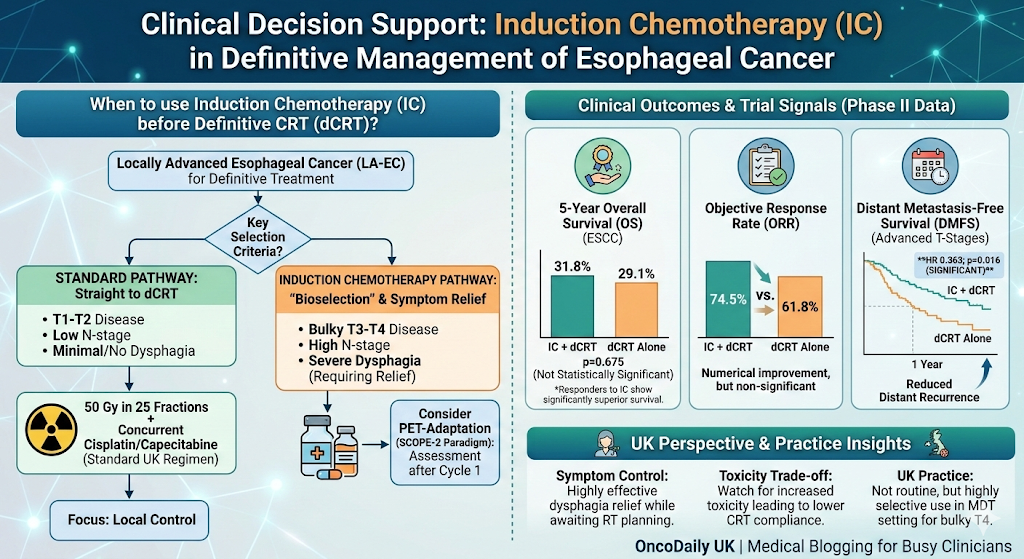
ESMO Oesophageal CPG Interim
Update confirms perioperative FLOT as the
preferred standard over neoadjuvant chemoradiotherapy (CROSS) for locally
advanced oesophageal/OGJ adenocarcinoma, based on the phase III ESOPEC trial
(mOS 66 vs 37 months; HR 0.70). Tislelizumab–ChT added for first-line advanced
oesophageal SCC.[1][2]
•
EMA CHMP positive opinion
for retifanlimab (Zynyz) + carboplatin–paclitaxel
in first-line advanced squamous cell anal cancer (SCAC), based on
POD1UM-303/InterAACT-2 mature OS data (mOS 32.8 vs 22.2 months; HR 0.75). NICE
technology appraisal [ID6482] is now underway — watch for NHS commissioning
implications.[3][4][5]
•
ESMO revised prostate cancer
diagnosis guideline mandates mpMRI before biopsy,
transperineal approach as standard, and PSA density ≥0.15 as trigger for biopsy
in MRI-negative patients — directly relevant to UK referral pathways.[6]
•
ESMO publishes first-ever
EBAI framework for AI-based biomarkers in oncology,
classifying them into three tiers (A/B/C) with defined validation requirements
— a landmark for digital pathology and AI-driven treatment selection.[7][8]
•
Triple KRAS pathway
inhibition (daraxonrasib + afatinib + SD36 PROTAC)
achieves complete tumour regression in preclinical PDAC models (CNIO, published
in PNAS); important conceptual advance but years from clinical
translation.[9][10]
Upper GI: Oesophageal &
Gastric
ESMO Oesophageal
CPG Interim Update (Published ESMO Open, 12 Feb 2025 — now prominently
featured)
This guideline
update, authored by Obermannová and Leong for the ESMO Guidelines Committee,
incorporates two major phase III trials:[11]
•
ESOPEC trial (Phase III; N=438): Perioperative FLOT vs preoperative CROSS CRT in
resectable oesophageal AC. At median follow-up of 55 months, mOS was 66
months (FLOT) vs 37 months (CRT) (HR 0.70; 95% CI 0.53–0.92; p=0.01).
Three-year PFS: 51.6% vs 35.0% (HR 0.66).[12]
•
New recommendation [I, A]: Patients with resectable, locally advanced oesophageal/OGJ AC
should receive perioperative FLOT. Neoadjuvant CRT is now reserved for
FLOT-unsuitable patients [I, C].
•
Advanced oesophageal SCC: Tislelizumab–ChT recommended first-line for PD-L1 TAP ≥5% [I, A;
ESMO-MCBS 4]. Second-line tislelizumab [I, A] or nivolumab [I, A] endorsed
after platinum–fluoropyrimidine failure.
NHS relevance: FLOT is already widely used in UK practice; this formalises it as
the unambiguous first-choice. Tislelizumab has EMA approval but NICE appraisal
status should be monitored.
SHR-1701 + CAPOX
in High-Risk Gastric/GEJ Cancer
Post-hoc analysis of
a phase III trial (N=551 high-risk subgroup with liver metastases, diffuse
histology, or peritoneal disease) reported mOS 14.4 vs 10.1 months (HR
0.62; 95% CI 0.49–0.78) favouring the PD-L1/TGF-β bifunctional fusion protein
SHR-1701 added to first-line CAPOX. Notably, liver metastases subgroup: mOS
16.8 vs 10.3 months (HR 0.46). The dual targeting of PD-L1 and TGF-β is a novel
mechanism, but SHR-1701 is not EMA/MHRA-approved; unclear positioning versus
established nivolumab + ChT regimens in HER2-negative GC/GEJC.[13]
Hepatobiliary & Pancreatic
KRAS-Targeted
Therapy in Pancreatic Cancer
Two streams of KRAS-related data were prominently covered in the
ESMO Daily Reporter this period:
1.
Preclinical triple KRAS
inhibition (CNIO study, PNAS): Daraxonrasib
(RAS-ON inhibitor) + afatinib (EGFR-TKI) + SD36 (STAT3 PROTAC degrader)
produced complete tumour regression in GEM models of PDAC, with mice remaining
tumour-free >200 days. The ESMO Daily Reporter cautioned that overlapping
toxicities and the historical failure of Hedgehog pathway inhibitors in PDAC
(which looked similarly promising preclinically) demand sober appraisal. Not
clinically actionable yet.[14][15]
2.
INCB161734 (KRAS G12D
inhibitor) — Phase I data from ASCO GI 2026
(January): Monotherapy ORR ~37%, DCR ~78% in heavily pretreated PDAC.
Combination cohorts with mFOLFIRINOX and gemcitabine/nab-paclitaxel showed
early safety signals. Additionally, ASP3082 (setidegrasib), a KRAS G12D degrader
combined with mFOLFIRINOX, showed ORR ~58% in evaluable patients.[16][17]
NHS relevance: No immediate practice
change, but KRAS G12D-directed approaches are moving rapidly toward
registrational studies; molecular profiling for KRAS variant subtype is
increasingly warranted at MDT.
Biliary Tract
Cancer — No New Major Updates This Week
The ESMO biliary tract CPG was updated in late 2024 with
recommendations for first-line durvalumab + gemcitabine–cisplatin and
second-line targeted therapies (FGFR inhibitors, IDH1 inhibitors). No new data
of note in the 8–15 February window.[18]
Genitourinary
ESMO Prostate
Cancer Guideline Revision (Published January 2026)
The revised ESMO CPG for localised prostate cancer introduces
several practice-changing diagnostic recommendations:[19][20]
•
mpMRI mandatory before
biopsy [I, B], with PI-RADS scoring guiding
decision-making
•
Transperineal biopsy now the recommended technique over transrectal approach [III, B],
citing infection rates of ~1.2% vs up to 4.1% with TRUS
•
PSA density ≥0.15 ng/mL/cm³ triggers biopsy even with negative mpMRI
•
Risk-stratified early detection
in four defined groups (not population-based screening)
NHS relevance: Directly aligns with NHS
prostate diagnostic pathway reforms. Transperineal biopsy services will need to
be resourced across all trusts; some may require investment in local
anaesthetic transperineal biopsy capacity.
Prostate Biopsy:
Prospective Validation of Index Lesion-Focused iSB
Presented at ESMO Asia 2025 and featured on the ESMO Daily Reporter:
a multicentre prospective trial (N=564, biopsy-naïve men) demonstrated non-inferiority
of targeted biopsy + ipsilateral systematic biopsy (iSB) versus standard
12-core SB. This supports reducing unnecessary contralateral cores and may
decrease overdetection of indolent cancer — consistent with the guideline
trajectory above.[21]
Bladder &
RCC — Ongoing ESMO Educational Activity
ESMO is running the Preceptorship on Metastatic Bladder and
Kidney Cancer (18–19 Feb 2026, Lugano) and a Deep Dive webinar on RCC
(April 2026). No new phase II/III trial results in these tumour sites were
published in the 8–15 February window. For reference, the landmark KEYNOTE-905/EV-303
(pembrolizumab + enfortumab vedotin in cisplatin-ineligible MIBC) and the EV-302
data continue to inform UK practice; NICE TA1097 for enfortumab vedotin +
pembrolizumab in first-line metastatic urothelial cancer was endorsed in
October 2025.[22][23][24]
Non-Melanoma Skin Cancer
EMA CHMP
Positive Opinion: Retifanlimab for SCAC (29 January 2026)
The CHMP recommended extending the indication for Zynyz
(retifanlimab) to first-line advanced squamous cell carcinoma of the anal
canal in combination with carboplatin–paclitaxel. Key data:[25][26]
•
POD1UM-303/InterAACT-2 (Phase III; N=308): mPFS 9.3 vs 7.4 months (HR 0.63; p=0.0006); ORR
56% vs 44%. Mature OS: 32.8 vs 22.2 months (HR 0.75; p=0.03);
crossover-adjusted benefit ~15 months.[27][28]
•
Already FDA-approved
(May 2025). If EC grants marketing authorisation, this becomes the first
immunotherapy regimen approved for advanced SCAC in Europe.
•
NICE STA [ID6482]: Stakeholder list finalised January 2026; appraisal is in
development. Given orphan disease status and unmet need, an accelerated
timeline is plausible.[29][30]
NHS relevance: High priority — SCAC
patients currently managed with carboplatin–paclitaxel alone. Prepare MDT
protocols for retifanlimab integration pending NICE recommendation.
Cost-effectiveness data will be scrutinised given the rare disease setting.
Cross-Cutting: AI
& Digital Oncology
ESMO EBAI
Framework for AI-Based Biomarkers
The ESMO Precision
Oncology and Digital Health Task Forces published the ESMO Basic
Requirements for AI-based Biomarkers in Oncology (EBAI) in Annals of
Oncology. Key classification:[31][32]
|
Class |
Description |
Validation Required |
|
A |
AI quantification of established biomarkers (e.g., cell counting) |
Concordance studies |
|
B |
AI-based surrogate biomarkers (screening/enrichment) |
Analytical validation |
|
C1 |
Novel AI prognostic biomarkers |
Retrospective RWD/trial data |
|
C2 |
Novel AI predictive biomarkers |
Prospective clinical trial validation |
Three non-negotiable
requirements: clearly defined ground truth, demonstrated performance,
and proven generalisability across settings. This framework will be
critical for NHS adoption of AI pathology tools (e.g., Paige AI, PathAI) and
should inform local trust governance committees evaluating AI-driven
diagnostics.
Sources: ESMO
Daily Reporter, ESMO Open, Annals of Oncology, EMA CHMP opinions, NICE
technology appraisals, NEJM, The Lancet, PNAS. Content limited to items
published or prominently featured 8–15 February 2026 and closely preceding days
where directly relevant.
[1] https://pmc.ncbi.nlm.nih.gov/articles/PMC11889489/
[2] https://www.esmo.org/oncology-news/perioperative-chemotherapy-with-flot-improves-survival-among-patients-with-resectable-oesophageal-adenocarcinoma
[3] https://www.nice.org.uk/guidance/awaiting-development/gid-ta11625
[4]
https://www.ema.europa.eu/en/documents/smop/chmp-post-authorisation-summary-positive-opinion-zynyz-emavr0000247788_en.pdf
[5]
https://www.ema.europa.eu/en/news/first-immunotherapy-based-treatment-recommended-advanced-anal-cancer
[6]
https://www.medscape.com/viewarticle/esmo-revises-early-prostate-cancer-diagnosis-and-monitoring-2026a1000280
[7]
https://dailyreporter.esmo.org/spotlight/the-first-esmo-guidance-for-ai-based-biomarkers
[8] https://pubmed.ncbi.nlm.nih.gov/41260261/
[9]
https://www.chemdiv.com/company/media/pharma-news/2026/triple-kras-inhibition-in-pancreatic-cancer-preclinical-success-vs-clinical-reality/
[10]
https://dailyreporter.esmo.org/homepage/pancreatic-cancer/ras-inhibition-a-game-changer-in-pancreatic-cancer
[11] https://pmc.ncbi.nlm.nih.gov/articles/PMC11889489/
[12]
https://www.esmo.org/oncology-news/perioperative-chemotherapy-with-flot-improves-survival-among-patients-with-resectable-oesophageal-adenocarcinoma
[13]
https://dailyreporter.esmo.org/esmo-immuno-oncology-congress-2025/news/novel-combination-shows-survival-benefits-in-high-risk-gastric-gastro-oesophageal-junction-cancer
[14]
https://dailyreporter.esmo.org/homepage/pancreatic-cancer/ras-inhibition-a-game-changer-in-pancreatic-cancer
[15]
https://www.chemdiv.com/company/media/pharma-news/2026/triple-kras-inhibition-in-pancreatic-cancer-preclinical-success-vs-clinical-reality/
[16]
https://oncupdates.com/article/advances-in-pancreatic-and-biliary-tract-cancers-at-asco-gi-2026-expert-insights/
[17]
https://oncodaily.com/oncolibrary/top-pancreatic-cancer-updates-january2026
[18] https://pmc.ncbi.nlm.nih.gov/articles/PMC11846563/
[19]
https://www.cancer.fr/professionnels-de-sante/veille/nota-bene-cancer/bulletin-n-670-du-9-janvier-2026/local-and-locoregional-prostate-cancer-esmo-clinical-practice-guideline-for-diagnosis-treatment-and-follow-up
[20]
https://www.medscape.com/viewarticle/esmo-revises-early-prostate-cancer-diagnosis-and-monitoring-2026a1000280
[21]
https://dailyreporter.esmo.org/esmo-asia-congress-2025/esmo-asia-congress/study-provides-prospective-validation-of-an-alternative-biopsy-technique
[22]
https://oncodaily.com/organizer/european-society-for-medical-oncology-esmo
[23] https://www.esmo.org/meeting-calendar
[24]
https://www.health-ni.gov.uk/articles/nice-endorsed-technology-appraisals-20252026
[25]
https://www.ema.europa.eu/en/documents/smop/chmp-post-authorisation-summary-positive-opinion-zynyz-emavr0000247788_en.pdf
[26]
https://www.ema.europa.eu/en/news/first-immunotherapy-based-treatment-recommended-advanced-anal-cancer
[27]
https://dailyreporter.esmo.org/esmo-immuno-oncology-congress-2025/news/retifanlimab-prolongs-overall-survival-in-advanced-squamous-cell-anal-cancer
[28]
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(25)00631-2/fulltext
[29] https://www.nice.org.uk/guidance/gid-ta11625/documents/final-matrix
[30] https://www.nice.org.uk/guidance/awaiting-development/gid-ta11625
[31] https://pubmed.ncbi.nlm.nih.gov/41260261/
[32]
https://dailyreporter.esmo.org/spotlight/the-first-esmo-guidance-for-ai-based-biomarkers
[33] https://trial.medpath.com/drug/approvals/ema/4470cb465d053e2b
[34]
https://www.esmo.org/guidelines-news/advanced-and-metastatic-prostate-cancer-esmo-clinical-practice-guideline-for-diagnosis-treatment-and-follow-up
[35] https://www.sciencedirect.com/journal/annals-of-oncology/issues
[36] https://www.ncbi.nlm.nih.gov/books/NBK610429/
[37] https://www.esmo.org/newsroom
[38]
https://www.esmo.org/content/download/77789/1426712/file/ESMO-Clinical-Practice-Guidelines-Standard-Operating-Procedures.pdf
[39] https://esmo-2025.org
[40]
https://cromospharma.com/esmo-2025-insights-from-the-floor-what-will-really-change-oncology-in-2026/
[41] https://www.esmo.org/living-guidelines
[42] https://oncodaily.com/event/esmo-329848
[43] https://www.sciencedirect.com/science/article/pii/S0923753420424603
[44] https://www.youtube.com/watch?v=a7Q290e2EwQ
[45] https://www.wcrp-esmo.org/outreach
[46] https://pubmed.ncbi.nlm.nih.gov/39986705/
[47] https://www.esmo.org/oncology-news
[48] https://dailyreporter.esmo.org
[49] https://www.esmo.org/guidelines/guidelines-news
[50]
https://www.merck.com/news/merck-advances-oncology-innovation-highlighting-progress-in-new-tumor-types-and-earlier-stages-of-disease-at-esmo-2025/
[51]
https://www.ajmc.com/publications/evidence-based-oncology/february-2026
[52]
https://www.esmo.org/guidelines/guidelines-by-topic/esmo-clinical-practice-guidelines-genitourinary-cancers
[53]
https://www.mskcc.org/news/esmo-2025-msk-presents-new-research-including-latest-on-lung-and-pancreatic-cancer-treatments
[54]
https://www.esmo.org/about-esmo/discover-esmo-journals/annals-of-oncology
[55] https://pmc.ncbi.nlm.nih.gov/articles/PMC10937212/
[56] https://www.unboundmedicine.com/medline/journal/Annals_of_Oncology
[57] https://www.sciencedirect.com/journal/annals-of-oncology
[58] https://www.esmo.org/guidelines/esmo-clinical-practice-guidelines-genitourinary-cancers
[59]
https://www.sciencedirect.com/journal/annals-of-oncology/vol/37/issue/2
[60] https://www.esmo.org/guidelines/esmo-pocket-guidelines-mobile-app
[61] https://dailyreporter.esmo.org/news
[62]
https://www.urologytimes.com/view/news-network-prostate-cancer-and-bladder-cancer-at-esmo-2025
[63]
https://dailyreporter.esmo.org/all_editions?search%5Bareas%5D%5B0%5D=786
[64]
https://digestivecancers.eu/spotlight-on-progress-in-pancreatic-cancer-highlights-from-esmo-2025/
[65] https://www.urologytimes.com/conference/esmo-annual-congress
[66] https://www.oncodeva.net/upload/esmo-guidelines/esofag.pdf
[67]
https://www.esmo.org/oncology-news/ema-recommends-extension-of-therapeutic-indications-for-retifanlimab
[68] https://pmc.ncbi.nlm.nih.gov/articles/PMC12836566/
[69]
https://www.esmo.org/guidelines/guidelines-by-topic/esmo-clinical-practice-guidelines-gastrointestinal-cancers/oesophageal-cancer
[70]
https://www.esmo.org/guidelines/esmo-clinical-practice-guideline-oesophageal-cancer
[71]
https://ascopost.com/news/september-2024/retifanlimab-plus-chemotherapy-may-extend-progression-free-survival-in-patients-with-squamous-cell-anal-carcinoma/
[72]
https://www.oncnursingnews.com/view/novel-combination-mitigates-chemo-induced-myelosuppression-in-gastric-gej-cancer
[73]
https://ascopost.com/news/june-2025/addition-of-retifanlimab-to-first-line-chemotherapy-in-advanced-squamous-cell-carcinoma-of-the-anal-canal/
[74]
https://www.cancernetwork.com/view/novel-combo-suppressed-chemo-associated-myelosuppression-in-gastric-gej-cancer
[75]
https://www.mou.cz/esmo-clinical-practice-guideline-oesophageal-cancer/t1838
[76]
https://dailyreporter.esmo.org/esmo-congress-2024/gastrointestinal-cancers/addition-of-immunotherapy-to-chemotherapy-improves-progression-free-survival-in-anal-canal
[77] https://ascopubs.org/doi/10.1200/JCO.2025.43.4_suppl.335
[78] https://pubmed.ncbi.nlm.nih.gov/40517007/
[79]
https://www.onclive.com/view/frontline-shr-1701-plus-capox-delivers-superior-os-vs-capox-alone-in-gastric-gej-cancer
[80]
https://www.medicinesresources.nhs.uk/european-medicines-agency-validates-marketing-authorisation-application-for-retifanlimab-for-the-treatment-of-squamous-cell-anal-carcinoma-scac-1.html
[81]
https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-retifanlimab-dlwr-carboplatin-and-paclitaxel-and-single-agent-squamous-cell-carcinoma
[82] https://www.premera.com/medicalpolicies/15.01.045.pdf
[83]
https://apnews.com/press-release/business-wire/incyte-announces-positive-chmp-opinion-for-zynyz-retifanlimab-for-first-line-treatment-of-advanced-squamous-cell-carcinoma-of-the-anal-canal-scac-f93bcda7861e4bfdb4e72e39c603b8de
[84]
https://www.discoveriesinhealthpolicy.com/2025/12/from-esmo-requirements-for-ai-based.html
[85]
https://uro-med.ir/prostate-cancer/wp-content/uploads/2025/05/PIIS0923753420398987.pdf
[86]
https://patientworthy.com/2026/02/05/ema-endorses-first-immunotherapy-option-for-advanced-anal-cancer/
[87] https://digitalhealth.tu-dresden.de/new-international-framework-defines-standards-for-ai-based-biomarkers-in-oncology-ebai/
[88] https://pmc.ncbi.nlm.nih.gov/articles/PMC11321723/
[89]
https://oncpracticemanagement.com/issues/2025/august-2025-vol-15-no-8/fda-officials-approve-retifanlimab-dlwr-with-carboplatin-and-paclitaxel-and-as-single-agent-for-squamous-cell-carcinoma-of-anal-canal
[90]
https://www.cancer.fr/professionnels-de-sante/veille/nota-bene-cancer/bulletin-n-666-du-24-novembre-2025/esmo-basic-requirements-for-ai-based-biomarkers-in-oncology-ebai
[91]
https://www.delveinsight.com/asco-conference/article/paxg-cassandra-trial
[92]
https://www.analcancerfoundation.org/news/fda-approve-retifanlimab-dwlr-zynyz-for-advanced-anal-cancer-what-you-should-know/
[93]
https://www.sherringford.org/post/triple-combination-therapy-induces-complete-pancreatic-cancer-regression
[94] https://digestivecancers.eu/promising-advances-in-pancreatic-cancer-asco-2025-highlights/
[95]
https://www.onclive.com/view/frontline-retifanlimab-approaches-eu-approval-for-advanced-squamous-cell-carcinoma-of-the-anal-canal
[96]
https://www.targetedonc.com/view/neoadjuvant-paxg-outperforms-mfolfirinox-in-resectable-and-borderline-resectable-pdac
[97] https://www.nice.org.uk/guidance/gid-ta11625/documents/html-content
[98]
https://dailyreporter.esmo.org/news/ras-inhibition-a-game-changer-in-pancreatic-cancer
[99] https://letswinpc.org/research/new-neoadjuvant-treatment-in-trials/
[100] https://letswinpc.org/research/research-asco-gi-2026/
[101]
https://www.oncologynewscentral.com/pancreatic-cancer/paxg-beats-mfolfirinox-in-pancreatic-cancer-for-event-free-survival
[102]
https://repository.icr.ac.uk/bitstream/handle/internal/4376/ESMO-ProstateCancer-Revised.pdf?sequence=5
[103] https://www.nature.com/articles/s41575-025-01153-w
[104] https://www.noah.bio/blog/asco-gi-2026
[105] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11190518/
[106]
https://www.geisinger.org/-/media/onegeisinger/files/policy-pdfs/rlcrp/g/mpa-g2007-prostate-biopsy-specimen-analysis.pdf
[107] https://www.youtube.com/watch?v=sIphDKL1rx4
[108]
https://oncodaily.com/oncolibrary/liver-and-pancreatic-cancer-trials-esmo25
[109] https://uroweb.org/guidelines/prostate-cancer
[110]
https://www.esmo.org/guidelines/esmo-clinical-practice-guideline-biliary-tract-cancer
[111]
https://www.esmo.org/oncology-news/ema-recommends-granting-a-marketing-authorisation-for-retifanlimab
[112] https://scholarx.skku.edu/journal/2382
[113] https://www.ema.europa.eu/en/medicines/human/EPAR/zynyz