The New Dawn in Bladder Cancer: EV-302 and the End of the “Chemo-Only” Era
The New Dawn in Bladder Cancer: EV-302 and the End of the “Chemo-Only” Era
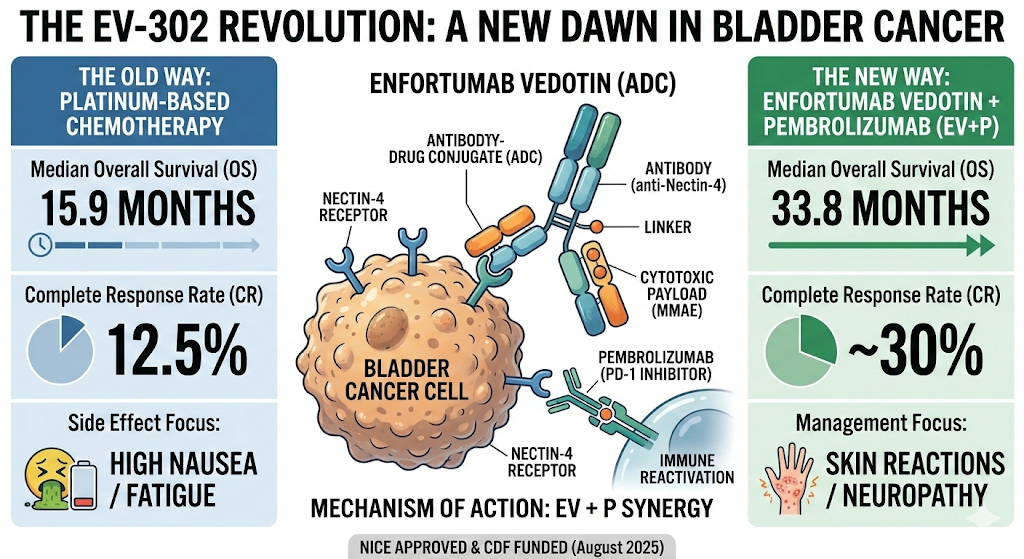
In a Nutshell: The standard of care for advanced urothelial cancer has remained stagnant for decades—until now. The combination of Enfortumab Vedotin and Pembrolizumab is nearly doubling survival rates, fundamentally rewriting the NHS treatment pathway.
🚀 Top 10 Clinical Takeaways
-
Survival Doubled: Median Overall Survival (OS) reached 33.8 months with the EV+Pembro combination vs. 15.9 months with standard chemotherapy (HR 0.51).
-
PFS Breakthrough: Progression-Free Survival (PFS) nearly doubled from 6.3 months to 12.5 months.
-
High Response Rates: The Objective Response Rate (ORR) was 67.7%, a staggering increase from the 44.4% seen with platinum-based chemo.
-
Complete Remissions: Nearly 30% of patients achieved a complete response (CR), compared to only 12.5% in the chemotherapy arm.
-
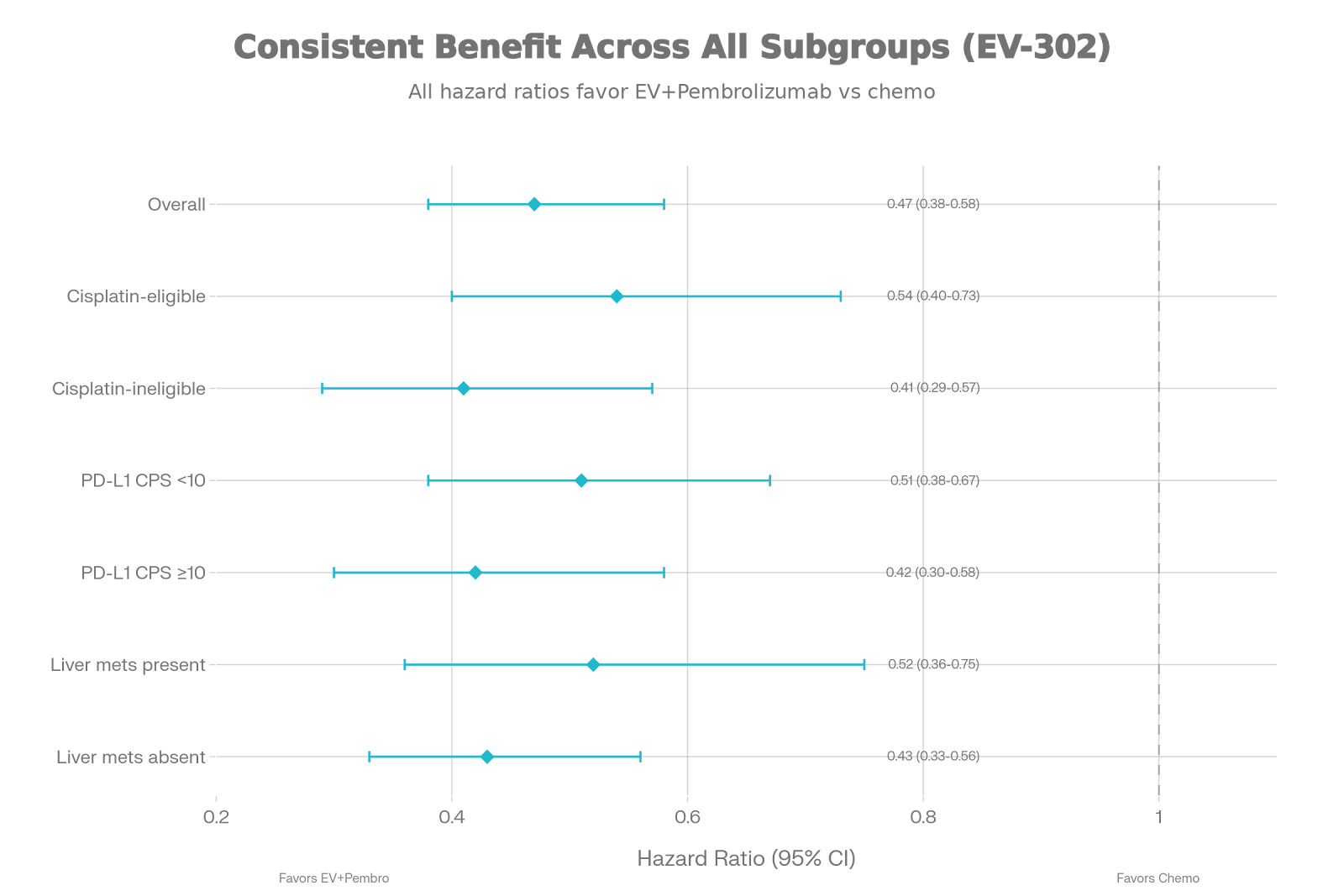
Cisplatin Status Irrelevant: The benefit was consistent regardless of whether patients were eligible for cisplatin or carboplatin.
-
Rapid Onset: Responses are often seen at the first scan (approx. 6–9 weeks), providing rapid symptomatic relief for patients with high disease burden.
-
Toxicity Profile: Key side effects include peripheral neuropathy, skin reactions, and hyperglycaemia; clinicians must be vigilant for “EV-related” dermatological toxicity.
-
NICE Approved: As of August 2025, NICE has recommended this combination for first-line use in the NHS for England and Wales.
-
Molecular Synergy: The combination pairs an Antibody-Drug Conjugate (EV) with a PD-1 inhibitor (Pembro), creating a “one-two punch” of targeted cell death and immune reactivation.
-
A New Standard: This replaces platinum-based chemotherapy as the preferred first-line option for metastatic urothelial carcinoma (mUC) in fit patients.
🔍 Deep Dive: The EV-302 / KEYNOTE-A39 Trial
The EV-302 trial is being hailed as the most significant advance in bladder cancer since the 1980s. For years, we relied on Gemcitabine and Cisplatin/Carboplatin, which, while effective initially, rarely led to long-term survival in the metastatic setting.
This global Phase III study randomized 886 patients with previously untreated locally advanced or metastatic urothelial cancer. The experimental arm received Enfortumab Vedotin (EV)—an ADC targeting Nectin-4—plus Pembrolizumab. The results showed a profound reduction in the risk of death by 49%.
The trial’s success lies in the synergy of its components. EV delivers a potent microtubule-disrupting agent directly to the cancer cells, while Pembrolizumab keeps the immune system’s T-cells from being “switched off.” This combination is particularly effective in bladder cancer, which is known for high Nectin-4 expression and a high tumor mutational burden.
🇬🇧 The UK Perspective: What This Means for Your Clinic
This is no longer a “trial-only” therapy; it is now an NHS reality.
-
NICE TA1097: Published in late 2025, this Appraisal officially brings EV+Pembro into the first-line setting.
-
The “Maintenance” Shift: Previously, the “UK Gold Standard” was chemotherapy followed by Avelumab maintenance (the JAVELIN Bladder 100 protocol). EV-302 largely supersedes this, as treating with the most potent combination upfront yields better OS than waiting for maintenance.
-
The Financials: The deal struck between NHS England, Astellas, and MSD ensures that despite the high cost, this combination is considered cost-effective for the 1,200+ eligible patients in the UK each year.
-
Logistics: Clinics will need to adapt to managing EV-specific toxicities. Unlike the familiar nausea of chemo, you’ll be looking for Maculopapular rashes and monitoring Hba1c more closely.
💡 Clinical Pearl
“If a patient is fit for therapy, EV+Pembro is no longer just an option; it is the benchmark. The 30% Complete Response rate is the number I now lead with in my patient consultations.”