Induction Chemotherapy in Esophageal Cancer: Is the “Head Start” Worth the Wait?
The “In a Nutshell” Intro
While definitive chemoradiotherapy (dCRT) is the gold standard for non-surgical esophageal cancer, systemic relapse remains a formidable foe for over 50% of patients. This post explores whether “bioselection” via induction chemotherapy (IC) can actually move the needle on survival or if it’s just delaying the inevitable.
Top 10 Clinical Takeaways
-
Distant Failure remains the Achilles’ heel: Over half of patients treated with dCRT alone fail distantly, not locally.
-
The Bioselection Signal: In the Sun Yat-sen Phase II trial, only responders to IC saw a survival benefit; unselected patients did not.
-
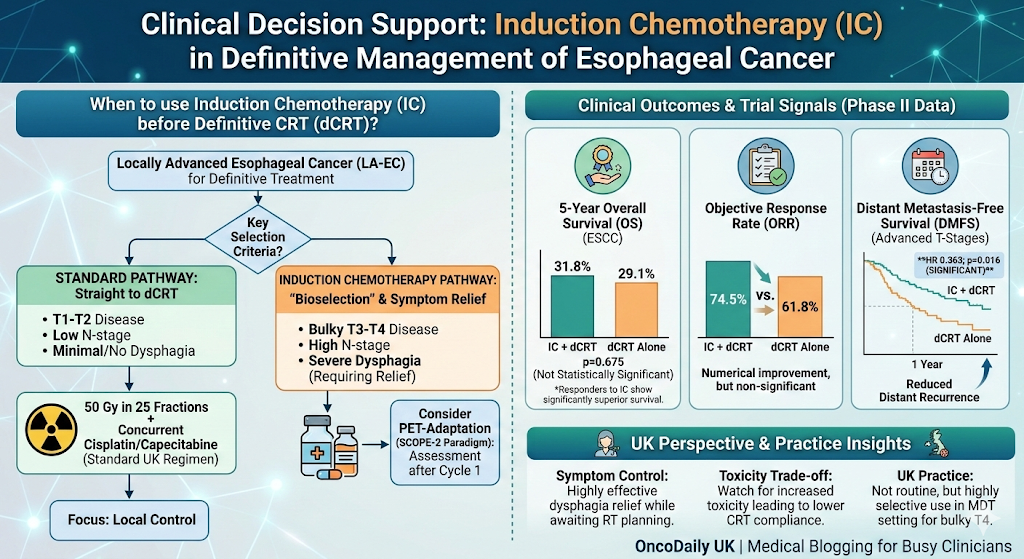
No Overall Survival (OS) “Magic Bullet”: 5-year OS in ESCC remains statistically similar between IC+dCRT (31.8%) and dCRT alone (29.1%).
-
DMFS Advantage: A 2025 multicenter study showed significant improvement in Distant Metastasis-Free Survival (HR 0.363; p=0.016) with IC.
-
T-Stage Matters: The greatest benefit for IC appears to be in advanced cT3–cT4 and TNM Stage III–IV disease.
-
Response-Adapted Strategy: Guidelines like ASCO suggest IC is only reasonable if using PET-response assessment (as seen in the CALGB 80803 paradigm).
-
UK Standard Dose: The RCR standard remains 50 Gy in 25 fractions (or 50.4 Gy in 28#).
-
The SCOPE-1 Legacy: Concurrent Cisplatin/Capecitabine remains the UK’s backbone for dCRT.
-
Symptom Control: IC is highly effective for rapid relief of dysphagia while awaiting complex RT planning.
-
The Toxicity Trade-off: Routine IC is not recommended because it may compromise a patient’s ability to complete the full course of radical RT.
Deep Dive Synthesis: Bioselection vs. Standard of Care
The logic behind induction chemotherapy (IC) is seductive: hit micrometastases early, shrink the target (GTV) to spare healthy lung and heart tissue, and test the tumor’s “mettle” before committing to a 5-week course of radiation. However, the academic landscape remains a sea of “non-significant” p-values.
The Sun Yat-sen Phase II Trial (focused on ESCC) is the most recent high-quality data point. While the Objective Response Rate (ORR) was numerically higher in the IC arm (74.5% vs 61.8%), this didn’t translate into a statistically significant 5-year OS benefit (p=0.675). The takeaway? If you use IC, you are effectively performing a “stress test” on the tumor. If the cancer responds, the patient likely has a favorable biology that will do well regardless; if it doesn’t, you may have just delayed effective local treatment.
More promisingly, a 2025 multicenter Western study highlighted that while OS didn’t jump, the Distant Metastasis-Free Survival (DMFS) did. This suggests that for those with bulky T3/T4 disease, IC might be the tool we need to prevent early systemic escape, even if it hasn’t yet reached the “Gold Standard” status in Phase III trials.
The UK Perspective: Tuesday Morning at the MDT
In the NHS, our practice is anchored by NICE (NG83) and the RCR fractionation guidelines. Currently, IC followed by dCRT is not a standard “off-the-shelf” pathway.
-
NICE/SMC Alignment: There is no specific mandate for IC. It is generally not funded as a routine precursor unless the patient is being treated within a clinical trial (like the ongoing SCOPE-2).
-
The “Clinic Reality”: If you have a patient with a bulky T4 tumor causing significant dysphagia and a 4-week wait for RT planning/mould room, IC (often FOLFOX or Cis/Cap) acts as a high-quality “bridge.”
-
The SANO Influence: With the SANO trial (2025) supporting active surveillance for those with a Clinical Complete Response (cCR), our focus in the UK is shifting toward organ preservation. If IC can help achieve that cCR, its role in the “Watch and Wait” pathway may expand.
Concluding Thought
“Induction chemotherapy is currently a tool for symptom control and bioselection, not a proven survival booster—use it for the bulky T4, but don’t delay radical RT for the T2.”
Suggested Infographic: The IC vs. dCRT Decision Matrix
Box Title: Clinical Decision Support for Induction Chemotherapy
-
Central Graphic: A flow-chart/decision tree.
-
Left Branch (Standard): T1-T2 disease -> Straight to dCRT (50Gy/25#) + Cis/Cap.
-
Right Branch (Consider IC): Bulky T3-T4, high N-stage, or severe dysphagia.
-
Key Stats Overlay: * ORR Improvement: ~12% (non-sig).
-
DMFS Benefit: HR 0.36 (Significant).
-
5-Year OS: ~30% in both groups.
-
-
Bottom Legend: “Follow SCOPE-2 PET-adaptation paradigm where available.”
Would you like me to draft a patient-facing summary of these findings to help with the consent process for induction chemotherapy?