Sorafenib in HCC
Sorafenib After Immunotherapy: Navigating Second-Line Treatment in Advanced HCC
A generation into the IO era, an old drug holds an evolving and contested place in the treatment sequence
For more than a decade, sorafenib stood alone at the frontier of systemic treatment for advanced hepatocellular carcinoma. Then came immunotherapy — and with it, a complete reordering of the treatment landscape. Atezolizumab–bevacizumab and durvalumab–tremelimumab are now the preferred first-line choices for most patients. Sorafenib, the former standard-bearer, has been repositioned: it is now a second-line option for patients who have progressed on these IO combinations.
This repositioning raises a question that guidelines answer only partially: how well does sorafenib actually work after IO, and for which patients is it the right choice?
What the Guidelines Say
The major hepatology and oncology bodies — AASLD, NCCN, ESMO, and ASCO — are aligned in listing sorafenib (alongside lenvatinib) as a preferred second-line TKI after first-line IO-based regimens in patients with Child-Pugh A liver function and ECOG performance status 0 or 1. That alignment, however, masks genuine uncertainty about the evidence base.
“The strength of recommendation for sorafenib in the post-IO setting is low — reflecting not doubt about the drug, but the absence of prospective randomised trials in this specific context.”
AASLD Guidance Commentary, 2024–26 (Weak recommendation, Level 5 evidence)
ESMO also lists sorafenib as a second-line option after first-line lenvatinib (Grade A, Level IV), though again the classification rests on non-randomised data and cross-trial comparisons rather than a dedicated RCT in that sequence.
The honest framing is this: sorafenib’s position in the second line is built on expert consensus and extrapolation from its historical first-line biology, not on prospective comparative post-IO data. That is not unusual in oncology, but it does shape how confidently clinicians can counsel patients.
The Evidence Foundation: SHARP and Beyond
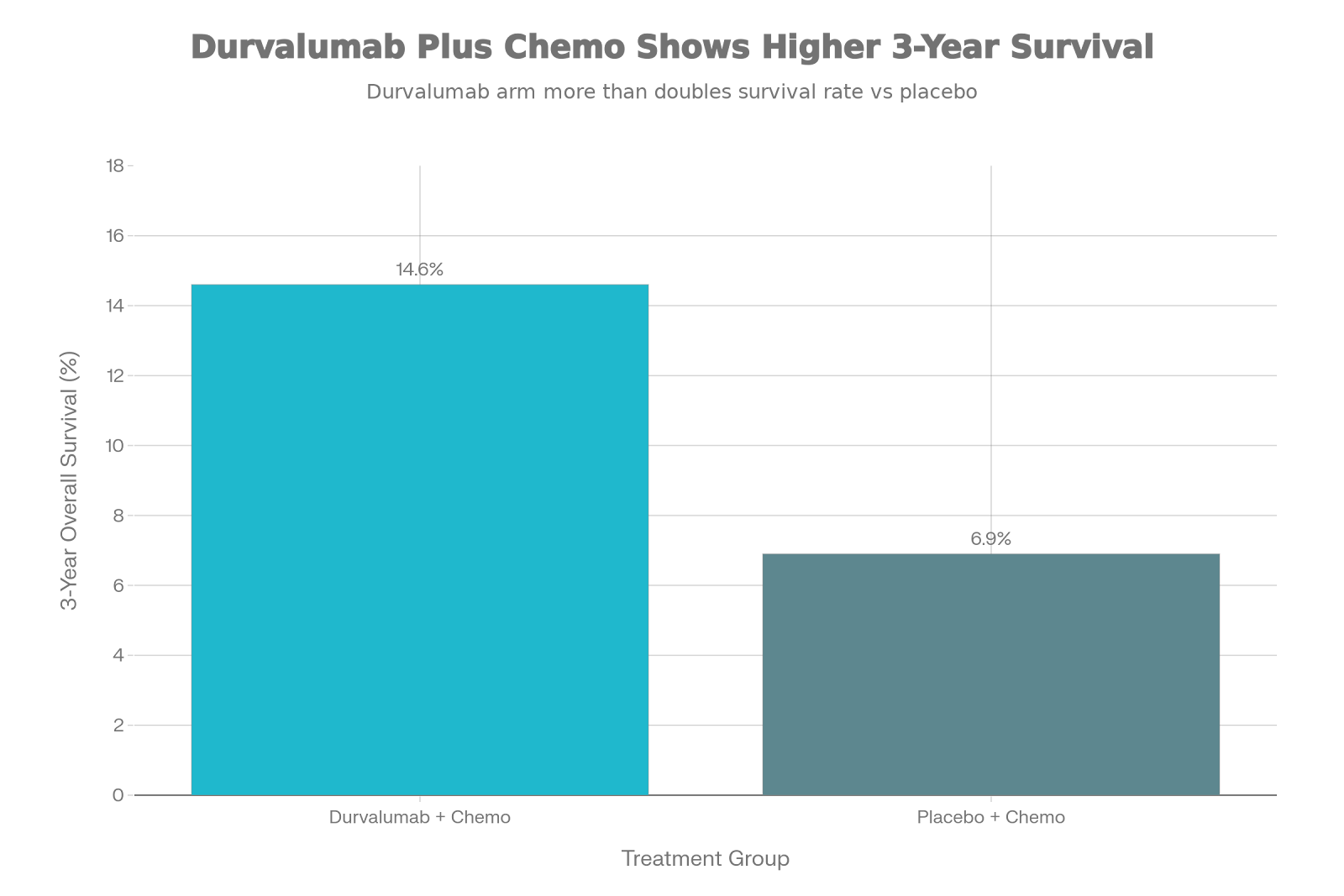
The data underpinning expectations of benefit when sorafenib is used second-line comes primarily from first-line trials — most importantly, the SHARP trial and its Asia-Pacific companion study — both of which enrolled TKI-naïve patients.
Key trial outcomes (first-line sorafenib vs placebo)
10.7 months
7.9 months
0.69 favours sorafenib
6.5 months
4.2 months
0.68 favours sorafenib
Meta-analyses across comparative TKI trials show sorafenib improves overall survival against sunitinib, brivanib, and linifanib, with a hazard ratio of approximately 0.8 in the broader advanced HCC population. Crucially, sorafenib appears to have a comparatively lower rate of severe adverse events than several newer TKIs — a practical advantage when selecting therapy for patients who may already carry IO-related immune toxicities.
Choosing Between Sorafenib and Lenvatinib
Both sorafenib and lenvatinib are guideline-listed for the second-line setting after IO, and the choice between them is not protocol-determined — it is patient-determined. Several factors favour one over the other in clinical practice.
Sorafenib tends to be favoured when patients have significant comorbidities (particularly hypertension or proteinuria, where lenvatinib’s profile may be less favourable), when there is a need for careful dose reduction in older or frailer patients who benefit from its established dose-titrability, and in Child-Pugh B7 patients where the longer track record of sorafenib at reduced doses provides a degree of reassurance.
Many centres also distinguish the sequence further: sorafenib and lenvatinib are preferred as the TKI of choice immediately after IO-IO or IO-VEGF first-line regimens, while cabozantinib and regorafenib are typically reserved for patients who have progressed on a prior TKI. This reflects their registration populations and the underlying clinical logic of the sequence rather than direct comparative data.
What to Expect: Outcome Expectations in TKI-Naïve Patients
For a patient with Child-Pugh A liver function and ECOG 0–1 who is progressing on first-line atezolizumab–bevacizumab or durvalumab–tremelimumab, the practical expectation from sorafenib second-line is modest but meaningful disease control, with an overall survival in the range of 8 to 12 months in TKI-naïve biology. These projections are extrapolated from first-line trial data and should be understood as approximations.
The absence of IO-sensitisation or resistance data makes it difficult to know whether prior IO exposure modifies the TKI response. This remains one of the most important unanswered questions in the field.
Managing Toxicity in the Second-Line Context
Sorafenib’s toxicity profile is well characterised. Hand-foot skin reaction is the signature adverse effect, occurring with sufficient frequency that prophylactic skin care regimens are now standard practice. Diarrhoea, fatigue, alopecia, and hypertension are the other commonly encountered toxicities. Bleeding risk is elevated relative to placebo, though grade 3–5 events are not clearly increased in systematic reviews — a relevant consideration in patients with portal hypertension or oesophageal varices.
The comparative severity advantage of sorafenib over some other TKIs may be particularly relevant in patients transitioning from IO, where residual immune-mediated toxicities (hepatitis, colitis, endocrinopathies) may already be present and compounding treatment tolerance.
Sorafenib in HCC: Treatment Pathway, Evidence & Patient Selection
Treatment sequence — advanced HCC (Child-Pugh A, ECOG 0–1)
line
IO-based combination (preferred)
Current guideline-preferred first-line systemic therapy for eligible patients
Durvalumab + tremelimumab
progression
Second-line TKI — sorafenib or lenvatinib ⬅ Focus of this review
Guideline-listed as preferred second-line TKIs in TKI-naïve patients. Choice driven by comorbidities, toxicity profile, and dose-titrability needs. Evidence base is extrapolated from first-line RCT data; no dedicated post-IO RCT exists.
Lenvatinib
line
Post-TKI options (after prior TKI)
Many centres favour cabozantinib or regorafenib after a TKI has been used. Registration populations for these agents were specifically TKI-pretreated patients.
Regorafenib
Ramucirumab (AFP ≥400)
Pivotal trial evidence — overall survival (sorafenib vs placebo, first-line)
Western
0.69
Eastern
0.68
vs TKIs
≈0.80
Recommendation strength — post-IO setting