MATTERHORN & SPOTLIGHT: Precision Medicine Redefining the Gastric Cancer Paradigm
MATTERHORN & SPOTLIGHT: Precision Medicine Redefining the Gastric Cancer Paradigm
In a nutshell: The “one-size-fits-all” FLOT era is evolving. With the MATTERHORN trial maturing and zolbetuximab entering the UK market, we are witnessing a shift toward biomarker-driven strategies in both the perioperative and metastatic settings.
Top 10 Clinical Takeaways: The New Gastric Standard
-
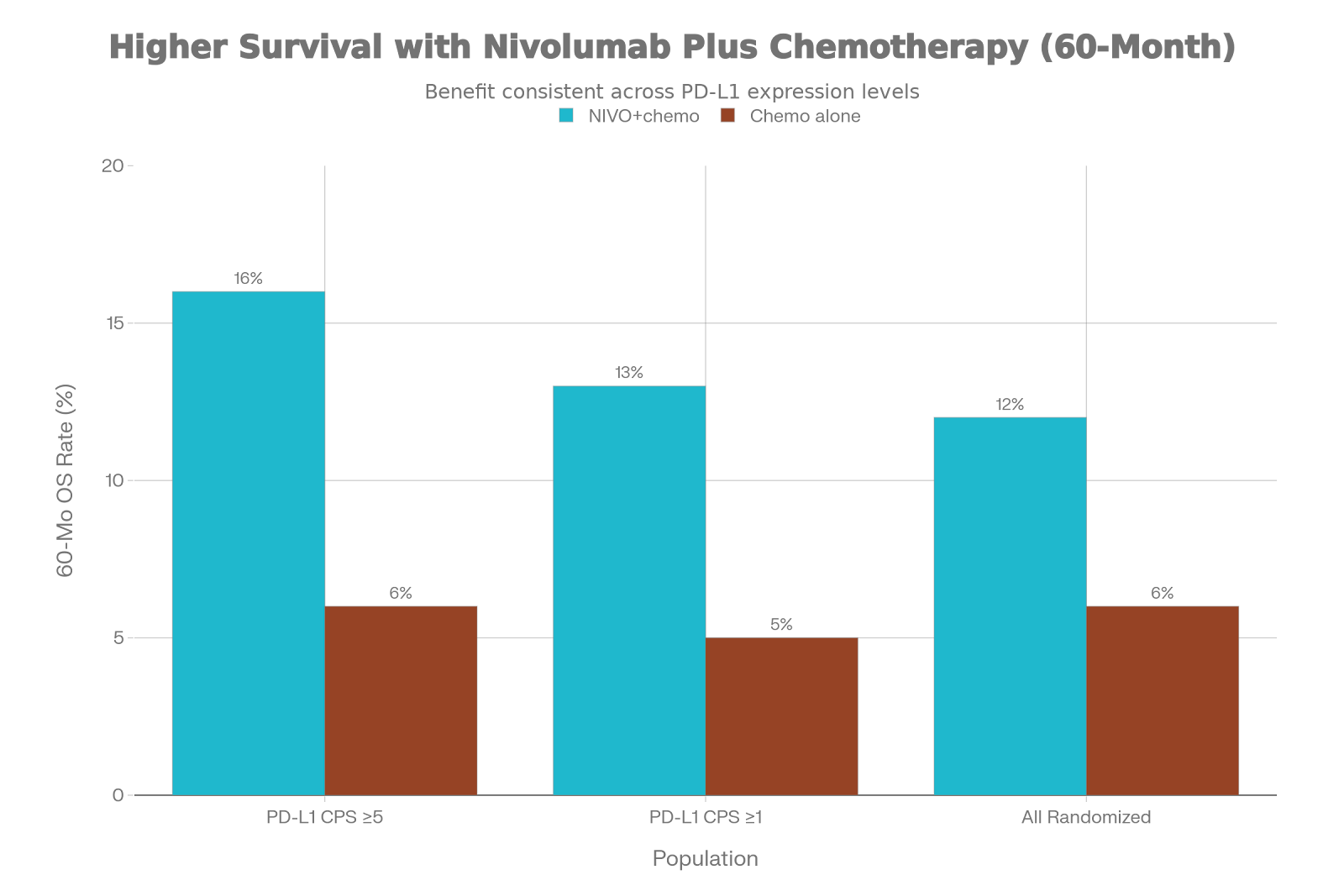
MATTERHORN OS Benefit: The addition of durvalumab to perioperative FLOT reduced the risk of death by 22% (HR 0.78, p=0.021) in the final 3-year analysis.
-
Pathological Milestone: In MATTERHORN, the pCR rate nearly tripled, rising from 7% (FLOT) to 19% (Durvalumab + FLOT).
-
PD-L1 Independence: Unlike the metastatic setting, MATTERHORN’s benefit was consistent regardless of PD-L1 status, supporting its use in a broader resectable population.
-
SPOTLIGHT OS Advantage: In the metastatic setting, zolbetuximab plus mFOLFOX6 extended median OS to 18.2 months vs 15.5 months for chemotherapy alone (HR 0.75).
-
The CLDN18.2 Biomarker: Zolbetuximab is only effective in patients with high expression (defined as ≥75% of tumour cells with 2+/3+ intensity).
-
Safety Profile (IO): Adding durvalumab to FLOT did not increase surgical complications or significantly worsen Grade 3/4 toxicity beyond expected immune-mediated AEs.
-
Safety Profile (Zolbetuximab): Nausea (82%) and vomiting (67%) are the primary “on-target” toxicities for zolbetuximab, requiring aggressive prophylactic anti-emetics.
-
Distant Metastasis Control: The MATTERHORN regimen significantly lowered distant recurrence rates, confirming early systemic micrometastatic clearance.
-
Nodal Clearance: A higher proportion of patients in the durvalumab arm achieved pN0 status (node-negative) at the time of surgery.
-
Testing Workflows: For a 2026 clinic, every new gastric biopsy now requires a “triple-threat” pathology request: HER2, PD-L1 (CPS), and CLDN18.2.
Deep Dive: From Perioperative Immunotherapy to Metastatic Precision
The MATTERHORN trial (n=948) has effectively set a new ceiling for resectable Gastric/GEJ adenocarcinoma. By integrating durvalumab into the standard 4+4 FLOT “sandwich” regimen, the trial addressed the high recurrence rates historically seen with chemotherapy alone. The doubling of pCR is the headline, but the 2-year EFS of 67.4% provides the clinical confidence needed for NHS adoption. Crucially, the “surgical window” was not compromised; R0 resection rates remained high (~87%), and there was no signal of increased perioperative mortality.
In parallel, for patients presenting with advanced disease, the SPOTLIGHT and GLOW trials have introduced zolbetuximab, a first-in-class monoclonal antibody targeting Claudin 18.2 (CLDN18.2). While immunotherapy (CheckMate-649) remains the backbone for PD-L1 high (CPS ≥5) patients, zolbetuximab offers a critical alternative for the ~38% of patients who are CLDN18.2-positive.
The clinical “read” is clear: We are moving toward a tiered approach. If resectable, we intensify with IO + FLOT. If metastatic, we branch by biomarker: Nivolumab for PD-L1 high, Zolbetuximab for CLDN18.2 high, and Trastuzumab (or T-DXd) for HER2 positive.
The UK Perspective: NICE, SMC, and Your Tuesday Clinic
The landscape in the UK has shifted rapidly in early 2026:
-
NHS Scotland Leading: The SMC has already granted “Interim Acceptance” for zolbetuximab in the metastatic setting, making it available for CLDN18.2+ patients in Scotland.
-
NICE Status: MATTERHORN (Durvalumab/FLOT) is currently under appraisal (ID6374). Until final guidance is published, FLOT-only remains the standard of care, but clinicians should be preparing for a transition to the “MATTERHORN protocol” via the Cancer Drugs Fund (CDF) later this year.
-
Biomarker Burden: The main challenge in a busy NHS lab is the volume of testing. CLDN18.2 testing (Ventana 43-14A assay) is being rolled out across regional Genomic Laboratory Hubs (GLHs).
-
Clinic Reality: For a patient fit for FLOT on a Tuesday morning, the conversation now involves explaining a longer treatment journey: neoadjuvant chemo-IO, surgery, adjuvant chemo-IO, followed by 10 cycles of durvalumab maintenance.
Concluding Thought
Clinical Pearl: The “Pathological Complete Response” in gastric cancer is no longer a rare win—with perioperative durvalumab, it is a nearly 1-in-5 reality that translates directly into long-term survival.