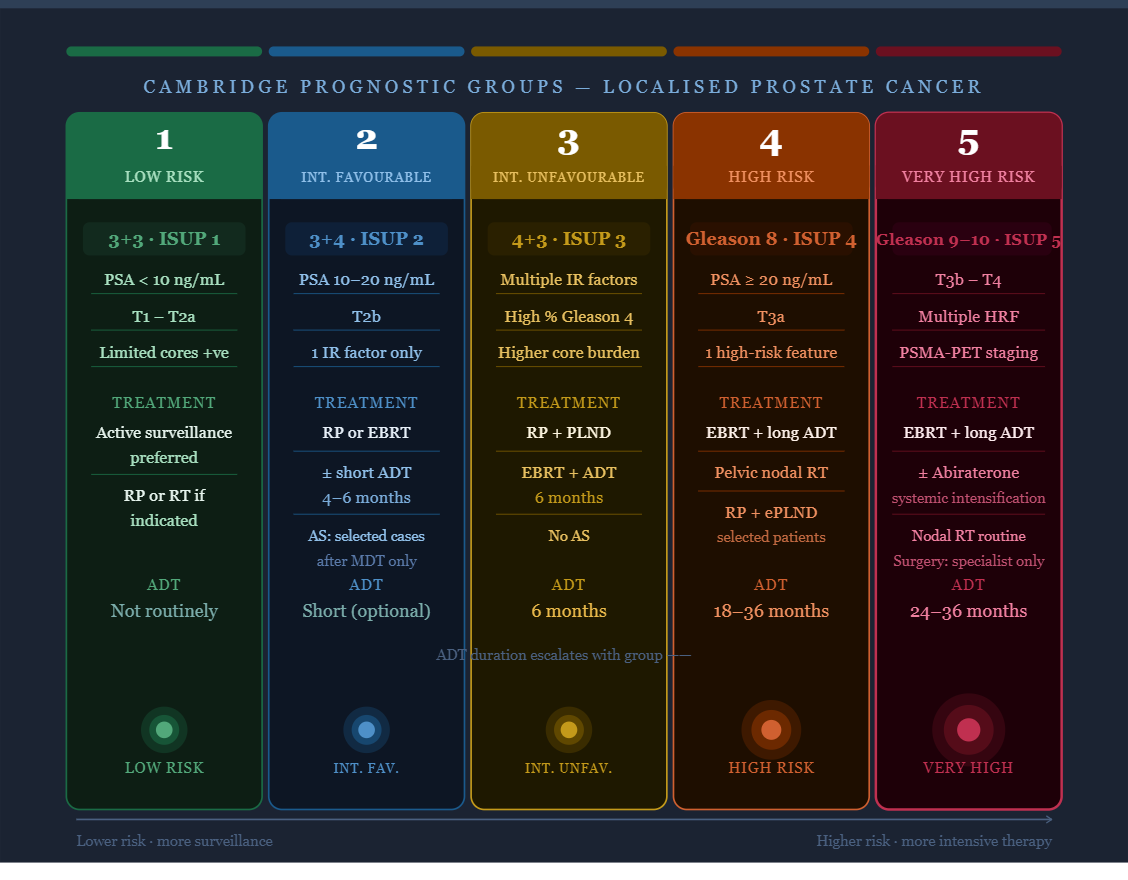
CPG Groups in Prostate cancer: Easy reference
Cambridge Prognostic Groups
CPG 1–5 at a glance
A fast-reference guide for busy clinicians — risk stratification, staging patterns, and treatment intent for localised prostate cancer
CPG 2 — Intermediate favourable
CPG 3 — Intermediate unfavourable
CPG 4 — High risk
CPG 5 — Very high risk
Group-by-group breakdown
Low risk
Gleason 3+3 (ISUP grade 1) · PSA <10 · T1–T2a · limited cores positive
Active surveillance or radical
ISUP 1
PSA <10 ng/mL
T1–T2a
≤50% cores positive
All features must be low-risk; a single unfavourable feature may upstage to CPG 2.
Active surveillance is preferred for eligible patients meeting NICE/local AS criteria.
Radical prostatectomy or radiotherapy (EBRT or brachytherapy) if patient preference, rapid PSA kinetics, or high-volume disease within CPG 1.
Intermediate — favourable
Gleason 3+4 (ISUP grade 2) · PSA 10–20 or T2b, otherwise low-risk features
RP or RT ± short ADT
ISUP 2
PSA 10–20 ng/mL
T2b
Only one intermediate-risk factor present. Predominantly 3+4 pattern; limited percentage of Gleason 4.
Radical prostatectomy or EBRT ± short-course ADT (4–6 months). Active surveillance may be considered in highly selected, well-informed patients after MDT discussion.
Intermediate — unfavourable
Gleason 4+3 (ISUP grade 3) · multiple intermediate-risk factors present
RP or RT + 6m ADT
ISUP 3
Multiple IR factors
High % Gleason 4
More than one intermediate-risk feature, or predominant Gleason 4 pattern. Higher recurrence risk than CPG 2.
Radical prostatectomy with pelvic lymph node dissection (if risk >5%).
EBRT + short-course ADT typically 6 months. Active surveillance not generally appropriate.
High risk
Gleason 8 (ISUP grade 4) · ≥T3 · PSA ≥20 (without very-high-risk features)
RT + long-course ADT
ISUP 4
PSA ≥20
T3a
One high-risk feature without the very-high-risk features of CPG 5 (Gleason 9–10, T3b–T4, multiple HRF).
EBRT + long-course ADT (18–36 months) is the standard. Radical prostatectomy with extended PLND in carefully selected patients. Pelvic nodal RT should be considered.
Very high risk
Gleason 9–10 (ISUP grade 5) · T4 · or multiple high-risk features
RT + long ADT ± systemic
ISUP 5
T3b–T4
≥2 high-risk features
Highest recurrence and mortality risk in localised/locally advanced disease. Staging with PSMA-PET strongly recommended before treatment planning.
EBRT + long-course ADT (24–36 months) ± abiraterone acetate intensification in fit patients.
Surgery in highly selected cases at specialist centres. Pelvic nodal irradiation routinely included.
Rapid-scan table
| CPG | Risk level | Gleason / ISUP | PSA | T-stage | Typical treatment intent |
|---|---|---|---|---|---|
| 1 | Low | 3+3 / ISUP 1 | <10 ng/mL | T1–T2a | Active surveillance or radical (RP/RT) |
| 2 | Intermediate — favourable | 3+4 / ISUP 2 | 10–20 ng/mL or | T2b | RP or RT ± short ADT (4–6 months) |
| 3 | Intermediate — unfavourable | 4+3 / ISUP 3 | Multiple IR factors | Multiple IR | RP or RT + ADT 6 months |
| 4 | High risk | Gleason 8 / ISUP 4 | ≥20 ng/mL or | ≥T3a | RT + long-course ADT (18–36m); RP in selected |
| 5 | Very high risk | Gleason 9–10 / ISUP 5 | Any (if multiple HRF) | T3b–T4 | RT + long ADT ± systemic intensification |
Important caveats
- Treatment intent varies with age, comorbidity, performance status, and patient preference — MDT discussion is essential for all CPG 3–5 cases
- CPG 2 vs CPG 3 boundary: the percentage of Gleason 4 pattern and number of positive cores are important discriminators beyond the simple Gleason score
- PSMA-PET/CT should be considered for CPG 4–5 to exclude occult metastatic disease before local treatment
- ADT duration guidance (6m, 18m, 36m) reflects broad NICE/EAU principles; institutional protocols and individual patient factors will modify this
- These groups apply to localised or locally advanced (non-metastatic) prostate cancer only
Source: Cancer Research UK — Cambridge Prognostic Groups. Treatment pathways based on NICE NG131 and EAU Guidelines on Prostate Cancer 2025. This reference is for healthcare professionals and is not a substitute for MDT discussion and individualised clinical decision-making.