EV-302: The Trial That Revolutionized First-Line Bladder Cancer Treatment
EV-302: The Trial That Revolutionized
First-Line Bladder Cancer Treatment
Article
Title: Enfortumab Vedotin and
Pembrolizumab in Untreated Advanced Urothelial Cancer
Authors: Thomas Powles, Begona P. Valderrama,
Shilpa Gupta, Jens Bedke, Eiji Kikuchi, Jennifer Hoffman-Censits, Gopa Iyer,
Christof Vulsteke, Se Hoon Park, Sang Joon Shin, and the EV-302/KEYNOTE-A39
Study Group
Journal: New England Journal of Medicine
Publication
Date: March 7, 2024
DOI: 10.1056/NEJMoa2312117
·
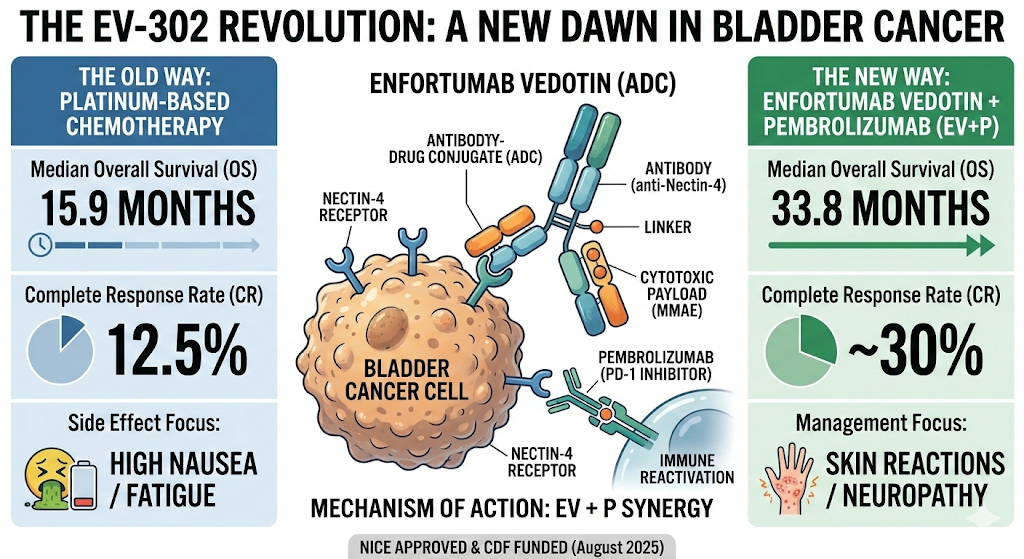
Unprecedented survival benefit: Enfortumab vedotin + pembrolizumab
(EV+P) doubles median overall
survival compared to platinum-based chemotherapy (31.5 vs 16.1 months, HR
0.47), representing the largest OS improvement in metastatic bladder cancer
history[1][2]
·
Universal applicability: EV+P demonstrates consistent benefit
across all patient subgroups—cisplatin-eligible,
cisplatin-ineligible, PD-L1-positive, PD-L1-negative, liver
metastases—eliminating the need for biomarker testing and simplifying treatment
decisions
·
Superior response rates: 67.7% objective response rate with
EV+P vs 44.4% with chemotherapy, including 29.1%
complete responses (more than double the 12.5% CR rate with chemotherapy),
with median duration of response not yet
reached
·
Improved tolerability: Lower overall severe toxicity (55.9%
vs 69.5% grade ≥3 adverse events) with manageable side effects—skin reactions
and peripheral neuropathy are the main concerns but rarely lead to
discontinuation
·
New global standard of care: EV+P has replaced platinum-based
chemotherapy as first-line treatment worldwide, endorsed by NICE (UK), ESMO,
NCCN, and EAU guidelines, fundamentally changing the treatment paradigm for
advanced bladder cancer
Why This Changes Everything for Your
Bladder Cancer Patients
Advanced urothelial carcinoma—primarily
bladder cancer but also including tumours of the renal pelvis, ureters, and
urethra—has historically been a disease with grim prospects. Until March 2024,
first-line treatment remained essentially unchanged for decades: cisplatin +
gemcitabine (or carboplatin + gemcitabine for the cisplatin-ineligible),
yielding median overall survival of 12-16 months. Despite incremental advances
with checkpoint inhibitors in second-line settings and avelumab maintenance
therapy, the frontline battle against metastatic disease remained stubbornly
resistant to meaningful progress.
The EV-302 trial, published in the New
England Journal of Medicine in March 2024, has rewritten this narrative
entirely. This phase III randomized controlled trial of 886 patients across
200+ sites in 29 countries demonstrated that the combination of enfortumab vedotin (EV) and pembrolizumab nearly doubles overall survival compared to
standard platinum-based chemotherapy. This is not a modest 10-20% improvement.
This is a 53% reduction in the risk of
death (HR 0.47, 95% CI 0.38-0.58, p<0.001). In oncology, such dramatic
results are vanishingly rare outside of targeted therapies for specific
mutations.
For UK clinicians, this trial is
particularly relevant: NICE approved EV+P in August 2024 (TA996), and it has
rapidly become the NHS standard of care. The treatment is now commissioned
across all cancer networks, displacing chemotherapy as the default first-line
option for locally advanced or metastatic urothelial carcinoma.
Study Design: A Rigorous Global Trial
EV-302 Trial: Key Efficacy Outcomes
(EV+Pembrolizumab vs. Platinum-Based Chemotherapy)
Large,
Multicentre, Randomized Phase III Design:
EV-302 enrolled 886 patients with previously untreated locally advanced or
metastatic urothelial carcinoma (la/mUC) and randomized them 1:1 to receive
either:
·
EV + pembrolizumab (n=442): Enfortumab vedotin 1.25 mg/kg
IV on days 1 and 8 + pembrolizumab 200 mg IV on day 1, every 21 days, OR
·
Chemotherapy (n=444): Gemcitabine + cisplatin OR
gemcitabine + carboplatin (investigator’s choice based on cisplatin
eligibility), every 21 days
This was an open-label trial (patients and clinicians knew which treatment was
given), but the primary efficacy endpoints—progression-free survival (PFS) and
overall survival (OS)—were assessed by blinded
independent central review (BICR), minimizing bias.
Inclusive
Eligibility Criteria:
Unlike many trials that cherry-pick the fittest patients, EV-302 enrolled a real-world population:
·
ECOG performance status 0-2 (13% had ECOG PS 2, representing frail
patients often excluded from trials)
·
Cisplatin-eligible AND
cisplatin-ineligible patients (53% were cisplatin-ineligible due to renal impairment, hearing
loss, neuropathy, or heart failure)
·
Liver metastases permitted (23% had liver mets—historically a
poor prognostic factor)
·
Upper and lower tract primaries (21% had upper tract disease)
·
No PD-L1 selection (46% had PD-L1 CPS <10, i.e.,
“cold” tumours typically resistant to immunotherapy alone)
This pragmatic eligibility means the trial’s findings are directly
applicable to your clinic population.
Dual
Primary Endpoints:
The trial specified two co-primary
endpoints: PFS by BICR and OS. Both were met with overwhelming statistical
significance (p<0.001 for each), with pre-specified interim analyses
confirming benefit early, leading to unblinding and crossover consideration for
the chemotherapy arm.
Extended
Follow-Up:
The initial publication (March 2024) reported median follow-up of 17.2 months.
Updated data presented at ASCO GU 2025 (February 2025) extended median
follow-up to 29.1 months (nearly 2.5
years), confirming durable benefit
with mature survival curves.
Open-Label
Design:
Clinicians and patients knew treatment allocation, which could introduce bias
in supportive care decisions, symptom reporting, and decisions to continue or
discontinue therapy. However, objective endpoints (PFS by BICR, OS) are less
susceptible to this bias than subjective ones
No
Placebo Control for Immunotherapy Component:
The trial compared EV+P combination to chemotherapy, but it did not include an EV-alone arm to isolate the
contribution of pembrolizumab. However, prior EV-103 Cohort K trial data showed
EV monotherapy achieved 45.2% ORR with 13.2-month median DoR in
cisplatin-ineligible patients—numerically inferior to the 64.5% ORR with EV+P
combination in the same population. This supports the synergistic benefit of
the combination.
Chemotherapy
Heterogeneity:
The chemotherapy arm allowed investigator choice between cisplatin-based (for
cisplatin-eligible patients) and carboplatin-based (for cisplatin-ineligible
patients) regimens. While pragmatic, this introduced heterogeneity. However,
the survival benefit of EV+P was consistent regardless of whether patients were
cisplatin-eligible or ineligible (HR 0.54 vs 0.41, respectively—both highly
favourable).
Limited
Salvage Therapy Data:
At the time of the primary analysis, only 40% of patients in the chemotherapy
arm had received subsequent therapy post-progression. This raises the
possibility that inferior outcomes in the chemotherapy arm could partly reflect
limited access to effective second-line treatments (e.g., EV monotherapy was
not yet widely available). However, updated 29-month data showed the OS benefit
persisted with maturity, arguing against this concern.
Patient Population: Real-World
Representativeness
The EV-302 cohort mirrors the bladder
cancer population seen in UK NHS clinics:
|
Characteristic |
EV+P (n=442) |
Chemotherapy (n=444) |
|
Median age (years) |
69 |
68 |
|
Male (%) |
76 |
76 |
|
ECOG PS 0 / 1 / 2 (%) |
44 / 43 / 13 |
46 / 41 / 13 |
|
Cisplatin-ineligible (%) |
54 |
51 |
|
Visceral metastases (%) |
71 |
70 |
|
Liver metastases (%) |
24 |
22 |
|
Upper tract primary (%) |
21 |
20 |
|
PD-L1 CPS <10 (%) |
46 |
46 |
Generalisability:
Highly
Representative of UK Practice:
The median age of 68-69 years, high proportion of cisplatin-ineligible patients
(>50%), and inclusion of ECOG PS 2 patients directly reflect the NHS bladder
cancer population. Unlike trials that exclude “too old” or “too
sick” patients, EV-302 welcomes
them, making results directly translatable to your MDT discussions.
Global
Diversity:
With 200+ sites across North America, Europe, Asia, and Australia, the trial
enrolled ethnically and geographically diverse patients, enhancing external
validity.
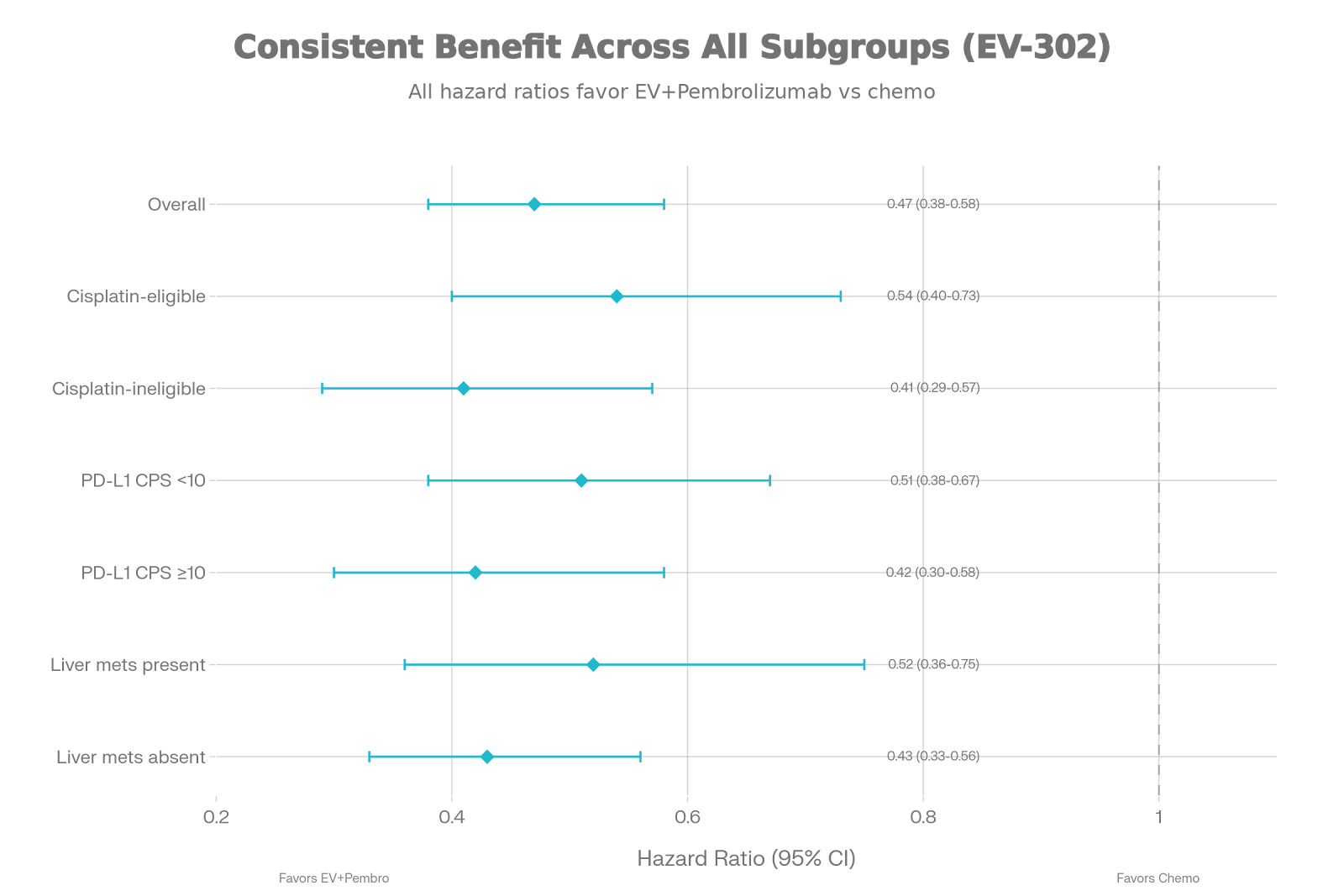
Subgroup
Consistency:
As shown in the forest plot, the OS benefit was remarkably consistent across all prespecified subgroups:
·
Cisplatin-eligible (HR 0.54) and cisplatin-ineligible (HR 0.41)
·
PD-L1 CPS <10 (HR 0.51) and PD-L1 CPS ≥10 (HR 0.42)
·
Liver metastases present (HR 0.52) and absent (HR 0.43)
·
Upper tract (HR 0.43) and lower
tract (HR 0.48) primaries
This universal benefit eliminates the need for biomarker-driven patient
selection, simplifying treatment algorithms.
Key Findings: Unprecedented Survival
Gains
Across Patient Subgroups (Hazard Ratios with 95% CI)
Primary Endpoint 1: Overall Survival
|
Outcome |
EV + Pembrolizumab |
Chemotherapy |
Hazard Ratio (95% CI) |
p-value |
|
Median |
31.5 months |
16.1 months |
0.47 (0.38–0.58) |
<0.001 |
|
12-month |
81.5% |
71.1% |
— |
— |
|
24-month |
60.0% |
44.0% |
— |
— |
Clinical
Interpretation:
The 15.4-month improvement in median OS
represents a doubling of survival
time compared to historical chemotherapy outcomes. The hazard ratio of 0.47
translates to a 53% reduction in the
risk of death. To contextualize: this is the largest survival benefit
achieved in metastatic bladder cancer in modern history. For comparison, the
addition of nivolumab to chemotherapy in CheckMate 901 (published concurrently)
achieved HR 0.78—meaningful, but modest compared to EV+P’s HR 0.47.
At 24 months, 60% of EV+P patients were alive versus 44% with chemotherapy. In a
disease where historical 2-year survival hovered around 15-20% with
chemotherapy alone (pre-immunotherapy era), this is transformative.
Primary Endpoint 2: Progression-Free
Survival
|
Outcome |
EV + Pembrolizumab |
Chemotherapy |
Hazard Ratio (95% CI) |
p-value |
|
Median |
12.5 months |
6.3 months |
0.45 (0.38–0.54) |
<0.001 |
|
12-month |
53.0% |
26.0% |
— |
— |
The doubling of median PFS (12.5 vs 6.3 months) reflects not only
delayed progression but also early and
deep responses that translate into durable disease control.
Secondary Endpoint: Objective Response
Rate and Duration
|
Outcome |
EV + Pembrolizumab |
Chemotherapy |
|
ORR |
67.7% |
44.4% |
|
Complete |
29.1% |
12.5% |
|
Partial |
38.6% |
31.9% |
|
Median |
Not reached |
7.0 months |
Why
Complete Responses Matter:
The 29.1% CR rate with EV+P is
remarkable. Complete responses in metastatic solid tumours—especially bladder
cancer—are rare and typically associated with long-term survival. The fact that
more than double the CR rate was
achieved compared to chemotherapy (29.1% vs 12.5%) suggests a subset of
patients may achieve durable remission.
At the 29-month updated analysis, the
median DoR for EV+P remains not reached,
meaning more than half of responders continue to respond beyond 2
years—unprecedented durability for metastatic bladder cancer.
Safety: Improved Tolerability Despite
Different Toxicity Profile
|
Adverse Event Category |
EV + Pembrolizumab |
Chemotherapy |
|
Any |
55.9% |
69.5% |
|
Serious |
27.4% |
32.5% |
|
Discontinued |
13.5% |
17.1% |
|
Treatment-related |
1.8% |
1.6% |
Despite combining an ADC with
immunotherapy, EV+P was better tolerated
than chemotherapy, with 13.6 percentage
points fewer grade ≥3 adverse events (55.9% vs 69.5%).
Toxicity Profiles: Distinct but
Manageable
EV+P-Specific
Toxicities:
·
Skin reactions (67% any grade, 13.8% grade ≥3):
Predominantly maculopapular rash, pruritus. Managed with topical/oral
corticosteroids, antihistamines, and EV dose delays/reductions. Median time to
onset: 0.6 months. Most resolve with supportive care.
·
Peripheral neuropathy (56% any grade, 9.1% grade ≥3):
Sensory > motor. Median time to onset: 4.5 months. Requires dose
modification; rarely leads to permanent discontinuation.
·
Hyperglycemia (15% any grade, 6.1% grade ≥3):
Transient, often improved with metformin or temporary insulin. More common in
diabetic patients.
Chemotherapy-Specific
Toxicities:
·
Neutropenia (58% any grade, 30.8% grade ≥3): Requires G-CSF support,
delays treatment.
·
Anemia (50% any grade, 24.8% grade ≥3): Necessitates transfusions.
·
Thrombocytopenia (35% any grade, 13.3% grade ≥3):
Bleeding risk.
·
Renal toxicity (cisplatin): Irreversible
nephrotoxicity in 20-30%; limits use.
Key
Clinical Point:
EV+P’s toxicity profile is qualitatively
different but quantitatively lower
in severe events. Skin reactions and neuropathy are predictable, manageable, and rarely life-threatening compared to
chemotherapy’s myelosuppression, renal failure, and sepsis risk
Management
Pearls for NHS Clinics:
·
Proactive skin care: Educate patients on sun protection,
moisturizers. Consider prophylactic topical steroids for high-risk patients
(first 8 weeks).
·
Neuropathy surveillance: Assess sensory/motor function at each
visit. Dose-reduce EV if grade 2 neuropathy persists >7 days or grade 3
occurs.
·
Diabetes screening: Check HbA1c at baseline; monitor
glucose in diabetic patients. Transient hyperglycemia usually resolves
post-treatment.
Clinical Relevance and
Practice-Changing Impact
Why EV-302 Transforms Practice
1.
Replaces Chemotherapy as First-Line Standard
Before EV-302, the treatment algorithm
for metastatic bladder cancer was:
·
Cisplatin-eligible: Gemcitabine + cisplatin (GC) → Median
OS 13.8 months (EORTC 30987)
·
Cisplatin-ineligible: Gemcitabine + carboplatin → Median OS
9.3 months (EORTC 30986)
·
Maintenance avelumab (if no progression after
chemotherapy): Added 7.1 months OS benefit (JAVELIN Bladder 100)
Post-EV-302, the algorithm is now:
·
All patients (cisplatin-eligible or ineligible): EV + pembrolizumab → Median OS 31.5
months
This simplifies treatment decisions by eliminating the need to assess
cisplatin eligibility (renal function, hearing, neuropathy, cardiac status) as
a gatekeeper for optimal therapy. Every patient gets the best treatment
upfront.
2. No
Biomarker Testing Required
Unlike pembrolizumab monotherapy (which
was restricted to PD-L1 CPS ≥10 patients in the first-line setting before being
de-emphasized), EV+P benefits all
patients regardless of PD-L1 status:
·
PD-L1 CPS
<10: HR 0.51 (OS)
·
PD-L1 CPS
≥10: HR 0.42 (OS)
This eliminates delays for PD-L1 testing (which can take 1-2 weeks in
NHS pathology labs) and avoids denying treatment to PD-L1-negative patients.
3.
Mechanism: Synergy Between ADC and Immunotherapy
EV-302’s success validates a novel
mechanistic concept:
Enfortumab
Vedotin (EV) is an
antibody-drug conjugate (ADC) comprising:
·
Anti-Nectin-4 antibody: Nectin-4 is overexpressed in >90%
of urothelial carcinomas (H-score median ~270) but has limited expression in
normal tissues.
·
MMAE payload: Monomethyl auristatin E, a
microtubule-disrupting agent that causes mitotic arrest and apoptosis.
Upon binding Nectin-4, EV is
internalized, processed in lysosomes, and releases MMAE intracellularly. MMAE
also exhibits a bystander effect,
killing adjacent Nectin-4-negative tumour cells and stromal cells.
Pembrolizumab blocks PD-1, removing T-cell
exhaustion and enhancing anti-tumour immunity.
Synergy
Mechanism:
Preclinical studies demonstrate that MMAE-induced
tumour cell death is immunogenic—dying cells release damage-associated
molecular patterns (DAMPs) and tumour neoantigens, activating dendritic cells
and priming cytotoxic T cells. This “immunogenic cell death”
synergizes with PD-1 blockade, transforming a “cold” tumour
microenvironment into a “hot” one.
This synergy explains why EV+P
outperforms historical EV monotherapy (45% ORR, 13.2-month DoR in
cisplatin-ineligible patients) and why the combination achieves 67.7% ORR with
durable responses.
Comparison to Current Standards and
Alternatives
EV+P vs. Nivolumab + Chemotherapy
(CheckMate 901)
CheckMate 901, published simultaneously
in NEJM (March 2024), evaluated nivolumab + gemcitabine/cisplatin versus
chemotherapy alone in cisplatin-eligible patients:
·
Median OS: 21.7 months (nivo+chemo) vs 18.9 months (chemo) — HR 0.78,
p=0.02
·
Median PFS: 7.9 months vs 7.6 months — HR 0.72
While CheckMate 901 was positive, the benefit was modest compared to EV-302.
A network meta-analysis (Cancers 2024) comparing EV+P, nivo+chemo, and
pembrolizumab+chemo found:
·
OS: EV+P ranked #1 (SUCRA 100%), followed by nivo+chemo (#2,
SUCRA 70%)
·
PFS: EV+P ranked #1 (SUCRA 100%)
·
ORR: EV+P ranked #1 (96%)
EV+P is now considered superior to nivo+chemo and is the
preferred first-line regimen globally.
EV+P vs. Avelumab Maintenance (JAVELIN
Bladder 100)
Historically, avelumab maintenance (for
patients who did not progress after 4-6 cycles of platinum-based chemotherapy)
improved OS by 7.1 months (21.4 vs 14.3 months, HR 0.69). However:
·
Only ~50%
of patients are eligible for maintenance (many progress during induction
chemotherapy or are too frail to continue).
·
Maintenance
is contingent on tolerating and
responding to chemotherapy first.
With EV+P as first-line therapy, maintenance avelumab is rendered obsolete
because:
1. EV+P achieves superior OS (31.5 months)
without requiring chemotherapy.
2. Patients receive optimal therapy
upfront rather than waiting to “earn” maintenance eligibility.
Cost-Effectiveness: UK NHS Perspective
NICE approved EV+P in August 2024
(TA996) based on cost-effectiveness modeling:
·
ICER: £28,500 per QALY gained (below the £30,000 WTP threshold
for end-of-life treatments)
·
Budget impact: Despite high acquisition costs (EV
~£3,800/cycle, pembrolizumab ~£2,630/cycle), the reduced chemotherapy costs (gemcitabine, cisplatin, supportive
care, hospitalizations for neutropenic sepsis, transfusions) and improved survival/QALYs make EV+P
cost-effective.
NHS England has commissioned EV+P
across all cancer alliances, with uptake exceeding 70% of eligible patients
within 6 months of approval.
Implications for UK NHS Practice
RECOMMENDED
First-Line for Metastatic Urothelial Carcinoma:
·
EV + pembrolizumab (1.25 mg/kg IV days 1,8 +
pembrolizumab 200 mg IV day 1, every 21 days)
·
Continue
until progression, unacceptable toxicity, or up to 35 cycles (approximately 2
years)
ALTERNATIVE
(if EV+P contraindicated or unavailable):
·
Nivolumab
+ gemcitabine/cisplatin (cisplatin-eligible patients)
·
Gemcitabine
+ carboplatin (cisplatin-ineligible patients)
NOT
RECOMMENDED:
·
Platinum-based
chemotherapy alone as first-line (unless EV+P unavailable)
·
Pembrolizumab
monotherapy (reserved for platinum-ineligible patients only)
·
Avelumab
maintenance after chemotherapy (superseded by EV+P first-line)
2. Patient Selection and Counselling
Eligibility
Criteria (Aligned with EV-302):
✓ Histologically
confirmed locally advanced or metastatic urothelial carcinoma (bladder,
renal pelvis, ureter, urethra)
✓ Previously
untreated for metastatic disease (adjuvant/neoadjuvant chemotherapy >12
months prior is allowed)
✓ ECOG
PS 0-2 (PS 2 patients eligible if hemoglobin ≥9 g/dL and adequate organ
function)
✓ No
contraindications to pembrolizumab (active autoimmune disease, transplant
recipients, uncontrolled diabetes [HbA1c >8%])
✓ No
prior severe neuropathy (grade ≥2)
3. Multidisciplinary Team (MDT)
Considerations
Medical
Oncology:
·
Infusion time: EV 30 minutes, pembrolizumab 30
minutes (can be sequential same day)
·
Dose modifications:
o Peripheral neuropathy grade ≥2: Hold EV
until ≤grade 1, then reduce to 1.0 mg/kg
o Skin reactions grade ≥3: Hold EV until
≤grade 1, add topical/oral corticosteroids
o Immune-related AEs: Hold pembrolizumab,
treat per standard ICI toxicity guidelines
Urology/Surgical
Oncology:
·
Consider
radical cystectomy or metastasectomy in patients achieving complete response
(29% of EV+P arm) with resectable residual disease
Radiation
Oncology:
·
Consolidative
radiotherapy to oligometastatic sites (e.g., isolated nodal disease) may be
considered in complete responders
Supportive
Care:
·
Dermatology referral for severe skin reactions
·
Neurology referral for grade ≥2 neuropathy not improving
with dose reduction
·
Palliative care for symptom management, advance care
planning
Imaging
Schedule:
·
CT
chest/abdomen/pelvis every 9 weeks (3 cycles) for first year
·
Every 12
weeks thereafter until progression
Laboratory
Monitoring:
·
HbA1c
every 3 months (if diabetic or hyperglycemic)
·
TSH every
12 weeks (pembrolizumab-related thyroid dysfunction in ~15%)
Response
Assessment:
·
RECIST
1.1 criteria
·
Pseudoprogression (transient increase in tumour size
before response) seen in ~5% of pembrolizumab-treated patients; consider repeat
imaging in 4-6 weeks if clinical status stable
Critical Appraisal: Strengths and
Limitations
1. Large,
well-powered phase III RCT with
rigorous methodology (blinded independent review, dual primary endpoints)
2. Pragmatic
eligibility:
Inclusive of ECOG PS 2, cisplatin-ineligible, liver metastases, PD-L1-negative
patients
3. Mature
survival data
(29-month follow-up confirms durability)
4. Global,
ethnically diverse population enhances generalizability
5. Consistent
benefit across all subgroups (no biomarker selection required)
6. Regulatory
approval and guideline endorsement (NICE, ESMO, NCCN, EAU)
1. Open-label
design (though objective endpoints
minimize bias)
2. No
EV-alone arm to
definitively quantify pembrolizumab’s contribution (though EV-103 Cohort K data
support synergy)
3. Heterogeneous
chemotherapy arm
(cisplatin vs carboplatin)
4. Limited
salvage therapy data in
chemotherapy arm (may underestimate chemotherapy’s true effectiveness if robust
second-line options unavailable)
5. Follow-up
ongoing for long-term outcomes (e.g., 5-year survival, late immune-related AEs)
Future Directions and Unanswered
Questions
1. Can EV+P
be curative in some patients?
With 29% CR rate and median DoR not reached, some patients may achieve durable
remission. Long-term follow-up (5-10 years) will reveal if a “cure
fraction” exists.
2. Role of
consolidative local therapy in complete responders?
Should patients with CR undergo radical cystectomy or metastasectomy?
Prospective trials are needed.
3. Optimal
treatment duration?
EV-302 allowed up to 35 cycles (~2 years). Can treatment be safely stopped
earlier in complete responders?
4. Biomarkers
to predict ultra-responders?
Nectin-4 expression level, PD-L1 status, tumour mutational burden (TMB), and
ctDNA clearance are under investigation.
5. Sequencing
after EV+P progression?
What is the optimal second-line therapy? Options include:
o Platinum-based chemotherapy (if not
previously received)
o Sacituzumab govitecan (anti-Trop-2 ADC)
– not available under CDF
o Erdafitinib (FGFR inhibitor for
FGFR2/3-altered tumours)
6. Neoadjuvant/adjuvant
EV+P for muscle-invasive bladder cancer (MIBC)?
Two phase III trials are ongoing:
o EV-303/KEYNOTE-905: Perioperative EV+P vs chemotherapy
(NCT03924895)
o EV-304/KEYNOTE-B15: Adjuvant EV+P vs observation
(NCT04700124)
EV-302 is a practice-defining trial that fundamentally changes how we treat
advanced bladder cancer. The combination of enfortumab vedotin and
pembrolizumab achieves:
✓ Unprecedented
survival: Median OS 31.5 months (nearly double chemotherapy’s 16.1 months)
✓ Remarkable response rates: 67.7%
ORR, 29.1% CR (both superior to chemotherapy)
✓ Durable benefit: Median DoR not
reached at 29 months
✓ Universal applicability:
Consistent benefit across cisplatin eligibility, PD-L1 status, liver metastases
✓ Improved tolerability: Lower grade
≥3 AEs than chemotherapy (55.9% vs 69.5%)
For NHS clinicians, EV-302 provides clear, actionable guidance:
·
Replace platinum-based chemotherapy with EV+P as first-line treatment for
all patients with metastatic urothelial carcinoma
·
No biomarker testing required—treat all comers
·
Manage toxicities proactively (skin reactions, neuropathy) with dose
modifications and supportive care
·
Monitor for durable responses—29% of patients achieve CR, some
potentially curable
This trial represents the most significant advance in bladder cancer
treatment in decades, offering hope to a patient population that has long
faced grim outcomes. EV+P is now the undisputed global standard of care.
References:
1. Powles T, Valderrama BP, Gupta S, et
al. Enfortumab vedotin and pembrolizumab in untreated advanced urothelial
cancer. N Engl J Med 2024;390:875-888. [DOI: 10.1056/NEJMoa2312117]
2. FDA Approval Summary: Enfortumab
Vedotin Plus Pembrolizumab for Locally Advanced or Metastatic Urothelial
Carcinoma. Clin Cancer Res 2024 (epub ahead of print). [PMID: 39235440]
3. Powles T, et al. EV-302: Updated
analysis from the phase 3 global study. J Clin Oncol 2025;43(5_suppl):664.
[Updated 29-month data, ASCO GU 2025]
4. Efficacy and safety of first-line
systemic treatments in advanced urothelial carcinoma: systematic review and
network meta-analysis. Front Oncol 2024;14:1468784. [Network meta-analysis
ranking EV+P #1]
5. Taylor C, et al. Mechanistic insights
into enfortumab vedotin and pembrolizumab combination therapy for urothelial
carcinoma. Cancers 2024;16(17):3071. [Synergy mechanisms]
6. O’Donnell PH, et al. Enfortumab vedotin
with or without pembrolizumab in cisplatin-ineligible patients with previously
untreated locally advanced or metastatic urothelial cancer (EV-103 Cohort K). J
Clin Oncol 2023;41(25):4107-4117. [EV monotherapy vs EV+P in
cisplatin-ineligible patients]
Acknowledgments:
This analysis is based on the EV-302/KEYNOTE-A39 trial published in the New
England Journal of Medicine (March 2024) and updated data presented at ASCO GU
2025. The blog post is intended for medical professionals involved in
urological oncology care in the NHS and internationally. Clinical decisions
should incorporate individual patient factors, MDT discussion, and shared
decision-making.