Practice Changing Article review : CheckMate 649 – Five-Year Survival Data Redefines the Standard of Care for Advanced Gastric Cancer
CheckMate
649: Five-Year Survival Data Redefines the Standard of Care for Advanced
Gastric Cancer
Article Citation
Title: Nivolumab (NIVO) + chemotherapy (chemo) vs chemo as first-line (1L)
treatment for advanced gastric cancer/gastroesophageal junction
cancer/esophageal adenocarcinoma (GC/GEJC/EAC): 5-year (y) follow-up results
from CheckMate 649
Authors: Markus H. Moehler, Jaffer A. Ajani, Lin Shen, Marcelo Garrido,
Carlos Gallardo, Lucjan Wyrwicz, Kensei Yamaguchi, James M. Cleary, Elena
Elimova, Ricardo Elias Bruges, Michalis Karamouzis, Tomasz Skoczylas, Arinilda
Bragagnoli, Tianshu Liu, Mustapha Tehfe, Stephen McCraith, Nan Hu, Jennifer
Zhang, Kohei Shitara
Journal: Journal of Clinical Oncology
Publication: January 27, 2025 (online); February 1, 2025 (print)
Volume/Issue: Volume 43, Number 4_suppl, Pages 398
Presentation: ASCO Gastrointestinal Cancers Symposium 2025
Trial Registry: NCT02872116
DOI: 10.1200/JCO.2025.43.4_suppl.398
5 Key Takeaway
Points
•
Nearly 3-fold improvement in 5-year survival: Nivolumab + chemotherapy
achieves 16% 5-year OS in PD-L1 CPS ≥5 patients versus only 6% with
chemotherapy alone
•
Durable benefit maintained over time: Hazard ratio remains 0.71 (29%
risk reduction) consistently from year 1 through year 5—no attenuation of
benefit
•
First landmark long-term data: This is the only gastric cancer
immunotherapy trial with published 5-year survival outcomes, establishing new
benchmarks for the field
•
No new safety signals at 60 months: The acceptable safety profile
documented in earlier analyses remains unchanged with extended follow-up
•
Validates NHS commissioning decisions: The 5-year data confirm NICE
TA857 approval of nivolumab + chemotherapy as standard first-line for PD-L1 CPS
≥5 advanced upper GI cancers
Background: Why This Matters
Gastric
cancer remains one of the deadliest malignancies globally, with over 1 million
new cases diagnosed annually. In the United Kingdom alone, approximately 15,700
new cases of gastric and oesophageal cancer are diagnosed each year.
The
Historical Reality: Before immunotherapy, the
prognosis for advanced gastric cancer was dismal:
•
Median overall survival: 8-11
months
•
2-year survival rate: 10-15%
•
5-year survival rate: <5%
For decades,
these outcomes remained essentially unchanged despite incremental improvements
in chemotherapy regimens. Then came CheckMate 649.
The CheckMate 649 Journey
When the
initial CheckMate 649 results were published in 2021, they demonstrated that
adding the anti-PD-1 antibody nivolumab to first-line chemotherapy could
improve overall survival in patients with high PD-L1 expression (CPS ≥5). This
led to regulatory approvals worldwide and NICE commissioning in the UK in
November 2022.
But a critical
question remained unanswered:
Would this
benefit persist long-term, or would outcomes eventually converge?
The 5-year
follow-up data, presented at ASCO GI 2025, provides the definitive answer: Yes,
and the benefit is even more striking than expected.
Study Design:
How Was This Research Conducted?
Patient Population
•
Total enrollment: 1,581 patients across multiple international sites
•
Inclusion criteria: Previously untreated, unresectable advanced/metastatic
gastric/GOJ/oesophageal adenocarcinoma
•
Key exclusion: HER2-positive tumors
•
Performance status: ECOG 0-1
only (excludes frail patients)
Treatment Arms
1.
Nivolumab + Chemotherapy (789 patients)
–
Nivolumab: 360 mg Q3W or 240 mg
Q2W
–
Chemotherapy: XELOX or FOLFOX
2.
Chemotherapy Alone (792 patients)
–
XELOX or FOLFOX
–
(A third arm with nivolumab +
ipilimumab was also studied but not highlighted in this update)
Outcomes Assessed
•
Primary: Overall Survival and Progression-Free Survival in PD-L1 CPS ≥5
patients
•
Secondary: OS/PFS in CPS ≥1 and all randomized populations, response rates,
duration of response
•
Safety: Adverse events, treatment discontinuations, late toxicities
Study Strengths
✓ Large sample size
(N=1,581) ✓ Long follow-up (minimum 60 months) ✓ Blinded independent review of
imaging ✓ Flexible chemotherapy backbone reflecting real-world practice ✓
Pragmatic eligibility criteria enhancing generalizability
Study Limitations
✗ Open-label design (no
blinding for patients/physicians) ✗ Abstract presentation (full manuscript
awaited) ✗ Limited detail on molecular subtypes (MSI status, EBV, etc.) ✗ Does
not include ECX regimen standard in some UK settings ✗ No detailed quality-of-life
reporting in this update
The Results:
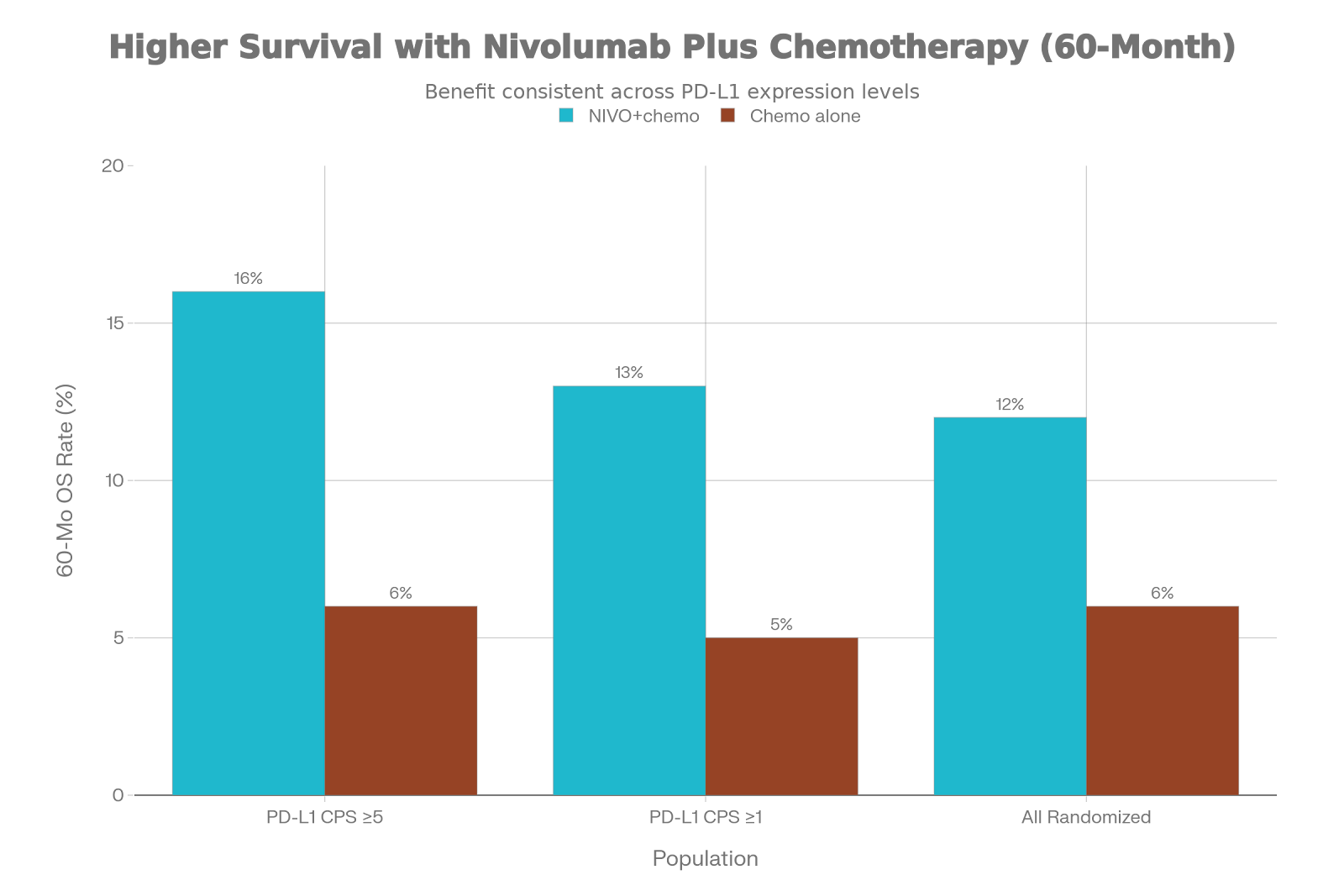
Transformative Long-Term Survival Data
5-Year Overall
Survival Rates
In PD-L1 CPS ≥5 Patients (Primary Population):
•
Nivolumab + Chemo: 16% alive at 5 years
•
Chemotherapy Alone: 6% alive at 5 years
•
Absolute Benefit: +10% (Number Needed to Treat = 10)
•
Hazard Ratio: 0.71 (95% CI 0.61-0.81) = 29% risk reduction in death
In PD-L1 CPS ≥1 Patients:
•
Nivolumab + Chemo: 13% alive at 5 years
•
Chemotherapy Alone: 5% alive at 5 years
•
Absolute Benefit: +8%
•
Hazard Ratio: 0.76 (95% CI 0.67-0.85)
In All Randomized Patients (Unselected):
•
Nivolumab + Chemo: 12% alive at 5 years
•
Chemotherapy Alone: 6% alive at 5 years
•
Absolute Benefit: +6%
•
Hazard Ratio: 0.79 (95% CI 0.71-0.88)
What This Means Clinically
The jump from
6% to 16% five-year survival in the CPS ≥5 population represents a nearly
3-fold improvement—a truly transformative finding for a disease
historically characterized by median survival of only 11 months.
Put simply:
•
Before CheckMate 649: 1 in 20 patients (5%) survived 5 years
•
After CheckMate 649: 1 in 6 patients (16%) survive 5 years
Other Key Efficacy Endpoints
Median
Overall Survival (mOS)
•
Nivolumab + Chemo: 14.4
months
•
Chemotherapy Alone: 11.1
months
•
Improvement: 3.3 months
Median
Progression-Free Survival (mPFS)
•
Nivolumab + Chemo: 8.3
months
•
Chemotherapy Alone: 6.1
months
•
Improvement: 2.2 months
Objective
Response Rate (ORR)
•
Nivolumab + Chemo: 60%
•
Chemotherapy Alone: 45%
•
Improvement: +15%
absolute benefit
Median
Duration of Response (mDOR)
•
Nivolumab + Chemo: 9.6
months
•
Chemotherapy Alone: 7.0
months
•
Improvement: 2.6 months
(more durable responses)
Important
Finding: The responses achieved with immunotherapy
are not only more frequent but also more durable, suggesting more stable
disease control.
The Survival Curve Story
When we examine
the actual survival curves over time, several striking patterns emerge:
1.
Early Benefit: The curves separate within 6 months, indicating that survival
advantage accrues early rather than slowly over years
2.
Persistent Separation: The curves maintain separation throughout the entire 60-month
period without converging—a sign of truly durable benefit
3.
The Plateau Effect: Around 36-48 months, the nivolumab + chemotherapy curve plateaus
at approximately 16-18%, suggesting a “cure fraction” where some patients
achieve long-term disease control
4.
Historical Context: Compared to pre-immunotherapy trials where <5% survived 5
years, the 16% rate represents an 8-fold improvement
Consistent Hazard
Ratios Across Time
One
of the most remarkable findings is the stability of hazard ratios:
•
Year 1 follow-up: HR = 0.71
•
Year 3 follow-up: HR = 0.71
•
Year 5 follow-up: HR = 0.71
This
consistency indicates that nivolumab + chemotherapy doesn’t simply delay death;
it fundamentally alters the disease trajectory for responders, creating
sustained long-term control that persists even after treatment discontinuation.
Safety Profile: What
About Toxicity?
The
abstract states “no new safety signals were identified” at 60-month
follow-up.
This
is reassuring because:
•
Checkpoint inhibitors can cause
delayed immune-related adverse events (irAEs) that emerge months or years after
treatment
•
Extended follow-up data are
critical for detecting rare late toxicities
From
Earlier CheckMate 649 Reports, We Know:
•
Grade 3-4 treatment-related
adverse events: 59% (nivo+chemo) vs 44% (chemo)
•
Discontinuation due to
toxicity: 20% vs 11%
•
Immune-related adverse events
(any grade): 32% vs 8%
•
Treatment-related mortality:
<1%
The
safety profile remains acceptable given the magnitude of survival benefit,
though the full manuscript will provide more detailed toxicity data.
Clinical
Relevance: Practice-Changing Implications
Why This Matters
for Patient Counseling
For
the first time, we have robust evidence-based estimates for long-term
survival expectations in advanced upper GI adenocarcinoma:
“If you have a PD-L1 CPS ≥5 tumor and tolerate nivolumab +
chemotherapy, approximately 1 in 6 patients (16%) will be alive at 5 years.”
This
fundamentally changes prognostic conversations and treatment planning.
Validation of
PD-L1 as a Predictive Biomarker
The
hierarchical testing strategy confirms that PD-L1 CPS ≥5 identifies the
population deriving the greatest benefit, justifying NICE’s restriction of
nivolumab commissioning to this biomarker-defined population.
However,
the substantial benefits seen in CPS ≥1 (HR 0.76) and even unselected patients
(HR 0.79) raise important questions about whether lower PD-L1 cutoffs might
also warrant immunotherapy.
Treatment Sequencing
Insights
The 5-year
data emphasize the importance of first-line delivery of the most effective
therapy. Why?
•
Patients have better
performance status at baseline
•
Greater treatment tolerance
early in disease course
•
Data consistently show that
metastatic patients derive more benefit from first-line than later-line
immunotherapy
Implications for UK NHS
Practice
Current Status
NICE Technology Appraisal TA857 (November
2022) approved nivolumab + chemotherapy for:
•
Untreated HER2-negative advanced gastric, GOJ, or oesophageal adenocarcinoma
•
PD-L1 CPS ≥5
The 5-year CheckMate 649 data validate this
decision. The actual 5-year survival outcomes meet
or exceed the projections used in NICE’s cost-effectiveness analysis.
Recommended Clinical
Pathway in NHS
Step 1: Diagnosis
& Staging
•
Confirm advanced/metastatic
gastric/GOJ/oesophageal adenocarcinoma
Step 2: HER2 Testing
•
If HER2-positive → Consider
trastuzumab-based pathway
•
If HER2-negative → Proceed to
Step 3
Step 3: PD-L1 Testing (Dako 28-8 assay)
•
CPS ≥5 → Nivolumab +
chemotherapy (Standard of care)
•
CPS 1-4 → Consider nivolumab +
chemotherapy (off-label) or chemotherapy alone
•
CPS <1 → Chemotherapy alone
or clinical trial
Step 4: Choose
Chemotherapy Backbone
•
FOLFOX
(fluorouracil/leucovorin/oxaliplatin)
•
CAPOX
(capecitabine/oxaliplatin)
•
ECX
(epirubicin/cisplatin/capecitabine) – if cisplatin-eligible
Step 5: Deliver
Nivolumab
•
6-8 cycles of chemotherapy
•
Nivolumab maintenance until
progression/toxicity (currently NICE approval for only 2 years in total)
•
Ongoing surveillance and
management of immune-related adverse events
Step 6: Long-Term
Survivorship
•
Surveillance imaging (CT every
3-6 months initially)
•
Monitor for late irAEs (thyroid
dysfunction, arthritis, etc.)
•
Psychosocial support and
rehabilitation
Implementation
Challenges & Solutions
|
Challenge |
Solution |
|
Timely PD-L1 |
Establish rapid |
|
Pathology expertise |
Training programs for |
|
Chemotherapy |
UK standard ECX |
|
Treatment duration |
Consensus |
|
Long-term |
Develop |
Comparison to
Historical Standards & Other Trials
Pre-Immunotherapy Era
|
Metric |
Historical Chemo |
CheckMate 649 |
|
Median OS |
9.9-11.2 months |
14.4 months |
|
2-Year OS |
10-15% |
~30% |
|
5-Year OS |
<5% |
16% |
|
HR Benefit |
– |
0.71 (29% |
Comparison to Other
Recent Trials
ATTRACTION-4 (Japan/South Korea)
•
Nivolumab + SOX vs SOX
•
Result: Did not meet primary
endpoint (mOS 17.5 vs 17.2)
•
Possible reasons: Asian
population, different chemotherapy backbone (S-1)
KEYNOTE-859 (Pembrolizumab)
•
Pembrolizumab + chemo vs chemo
•
Result: mOS 12.9 vs 11.5 months
(HR 0.78)—similar magnitude to CheckMate 649
KEYNOTE-062 (Pembrolizumab monotherapy)
•
Pembrolizumab alone vs chemo
•
Result: Pembrolizumab
monotherapy not recommended
CheckMate
649 Remains the Only Trial with Published 5-Year Data—Setting the Benchmark
Critical
Appraisal: Strengths & Limitations
Major Strengths
✓ Landmark long-term
data: First 5-year follow-up for anti-PD-1 + chemotherapy in upper GI
cancers
✓ Large robust sample:
N=1,581 with mature follow-up across multiple international sites
✓ Durable benefit:
Consistent hazard ratio (0.71) from year 1 through year 5—no attenuation
✓ Broad applicability:
Benefit across PD-L1 subgroups, geographic regions, and tumor subsites
✓ Quality-adjusted
survival: Earlier analyses confirmed that survival gains come with
acceptable quality of life (Q-TWiST analyses)
✓ Clinical practice
alignment: Directly informs NHS guidelines and commissioning policy
Important Limitations
✗ Abstract
format: Full manuscript not yet published—limited detail on subgroups,
detailed safety, and quality-of-life outcomes
✗ Open-label
design: Potential bias in supportive care, subsequent therapy selection,
and patient-reported outcomes
✗ Limited
molecular characterization: No reported outcomes by microsatellite
instability (MSI), Epstein-Barr virus (EBV), or genomic subtype
✗ Chemotherapy
backbone differences: UK standard (ECX) not directly represented;
extrapolation from FOLFOX/XELOX required
✗ Treatment
duration ambiguity: Abstract does not clarify optimal nivolumab duration or
proportion receiving maintenance therapy
Unmet Needs & Future
Directions
Despite
this landmark achievement, critical questions remain:
Current Research Gaps
1. PD-L1 CPS <5 Population
•
What is optimal first-line
therapy for CPS 1-4 or CPS 0 patients?
•
Should they receive nivolumab +
chemotherapy off-label, or continue with chemotherapy alone?
2. Perioperative Immunotherapy in
Earlier Disease
•
Can first-line immunotherapy
success translate to earlier-stage disease?
•
MATTERHORN Trial: Testing perioperative durvalumab + FLOT in resectable gastric
cancer
•
CheckMate 577: Adjuvant nivolumab after chemoradiotherapy for oesophageal cancer
3. Combination Strategies
•
HER2-positive disease: Can
immunotherapy be combined with trastuzumab?
•
Claudin 18.2-targeted therapy:
Can combination improve outcomes?
•
VEGF inhibition: Immunotherapy
+ ramucirumab synergy?
4. Biomarkers Beyond PD-L1
•
MSI status: Should MSI-high
tumors receive immunotherapy regardless of PD-L1?
•
EBV positivity: Are
EBV-associated gastric cancers uniquely immunogenic?
•
Tumor mutational burden: Can
TMB predict immunotherapy response?
5. Treatment Duration Optimization
•
How long should nivolumab
maintenance continue?
•
Can chemotherapy-free,
immunotherapy-only regimens work?
•
Can treatment be de-escalated
in sustained responders?
Quality of
Evidence Assessment (GRADE Methodology)
|
Domain |
Assessment |
Comments |
|
Study Design |
High |
Phase 3 RCT with appropriate randomization and stratification |
|
Sample Size |
High |
N=1,581 provides robust statistical power |
|
Follow-Up Duration |
High |
60-month minimum follow-up ensures mature data |
|
Blinding/Bias |
Moderate |
Open-label design; BICR mitigates but doesn’t eliminate bias |
|
Consistency |
High |
HR stable across populations and over time |
|
Directness |
High |
Directly addresses clinical question in intended population |
|
Precision |
High |
Narrow confidence intervals; events well-characterized |
|
Overall Quality |
HIGH |
Landmark trial providing definitive evidence for practice change |
The Bottom Line: What
Does This Mean?
For Patients
with Advanced Gastric/GOJ/Oesophageal Cancer
✓
If you have a PD-L1 CPS ≥5 tumor, nivolumab + chemotherapy offers a
realistic chance of long-term survival that would have been unthinkable a
decade ago.
✓
Approximately 1 in 6 patients (16%) with CPS ≥5 will survive ≥5
years—comparable to some solid tumors that are now considered “treatable.”
✓
Responses are not only more frequent (60% vs 45%) but also more durable (9.6 vs
7.0 months), suggesting more stable disease control and better quality of life
during treatment.
For NHS Oncologists & MDTs
✓ CheckMate 649
5-year data validate current NICE TA857 commissioning of nivolumab +
chemotherapy for CPS ≥5 patients.
✓ Standard
of care is confirmed: Nivolumab + chemotherapy (with PD-L1 CPS ≥5
selection) remains the benchmark for first-line treatment of HER2-negative
advanced gastric/GOJ/oesophageal adenocarcinoma.
✓ Long-term
survivorship pathways must be established to manage the growing cohort of
patients surviving ≥5 years.
For Health
Economists & Commissioners
✓
The 5-year OS benefit (16% vs 6%) meets or exceeds the projections used
in NICE’s cost-effectiveness analysis.
✓ Cost-effectiveness
ratios are likely more favorable than modeled; potential justification for
broader access or lower pricing thresholds.
✓ Budget
impact must account for extended nivolumab treatment duration and
subsequent therapy costs, but improved survival outcomes offset many downstream
expenses.
Clinical Pearls &
Practice Tips
For Oncologists
1.
Always test PD-L1: Universal testing in advanced upper GI adenocarcinoma should be
standard practice—it determines treatment eligibility.
2.
Discuss 5-year prospects: Use CheckMate 649 5-year data (16% OS in CPS ≥5) when counseling
patients—it reframes the conversation from palliative to potentially curative
intent.
3.
Maintain chemotherapy
intensity: All CheckMate 649 patients received
full-dose platinum-fluoropyrimidine doublets
4.
Plan for long-term follow-up: With 12-16% of patients surviving ≥5 years, survivorship clinics
and late toxicity monitoring become essential.
5.
Monitor for late irAEs: Immune-related adverse events can emerge months or years after
treatment cessation; remain vigilant for thyroid dysfunction, arthritis,
colitis, and endocrinopathies.
For Pathologists
1.
Standardize PD-L1 testing: Use Dako 28-8 assay (or validated equivalent) with robust quality
assurance.
2.
Distinguish CPS from TPS: Combined positive score (includes tumor and immune cells) differs
from tumor proportion score; ensure correct methodology.
3.
Provide timely reporting: Target <10 working days from specimen receipt to report to
minimize treatment delays.
4.
Document staining quality: Include controls, percentage positive cells, and interpreter
confidence to support clinical decision-making.
Looking Forward:
Ongoing Trials & Future Strategies
Perioperative Immunotherapy
MATTERHORN
Trial (NCT04592913)
•
Testing: Perioperative
durvalumab + FLOT vs FLOT alone
•
Population: Resectable
gastric/GOJ cancer
•
Timeline: Results expected
2025-2026
•
Hypothesis: Immunotherapy may
improve cure rates in earlier-stage disease
CheckMate
577
•
Testing: Adjuvant nivolumab vs
placebo after chemoradiotherapy
•
Population: Oesophageal/GOJ
cancer
•
Status: Ongoing; results in
development
Combination
Strategies Under Investigation
•
Dual checkpoint inhibition (anti-PD-1 + anti-CTLA-4)
•
Immunotherapy + VEGF
inhibition (nivolumab + ramucirumab)
•
Immunotherapy + HER2
targeting (pembrolizumab + trastuzumab +
chemotherapy)
•
Claudin 18.2-directed
therapy (zolbetuximab) + immunotherapy combinations
Biomarker Refinement
Initiatives
•
Circulating tumor DNA
(ctDNA): Liquid biopsies to guide treatment
duration
•
Spatial transcriptomics: Profiling tumor microenvironment architecture
•
Multi-omic integration: Combining PD-L1 + MSI + EBV + TMB + transcriptomics
•
Immune cell profiling: Characterizing TIL subset composition and exhaustion status
Disclaimer
This blog post summarizes
publicly available data from the CheckMate 649 trial and ASCO GI 2025
presentation for educational purposes. Treatment decisions should be made in
consultation with qualified healthcare professionals based on individual
patient circumstances and in accordance with NICE guidelines and NHS
commissioning policies.
References
[1] Moehler MH, et al.
Nivolumab + chemotherapy vs chemo as first-line treatment for advanced
GC/GEJC/EAC: 5-year follow-up from CheckMate 649. J Clin Oncol.
2025;43(4_suppl):398.
[2] CheckMate 649 4-year
follow-up analyses. ASCO GI 2024.
[3] CheckMate 649 3-year
follow-up results. J Clin Oncol. 2024.
[4] Q-TWiST analysis of
CheckMate 649. Gastric Cancer. 2025;28(1).
[5] Health-related quality of
life in CheckMate 649. J Clin Oncol. 2023;41(16_suppl).
[6] NICE Technology Appraisal
TA857: Nivolumab for advanced gastric cancer. November 2022.
[7] Summary of CheckMate 649
Chinese subgroup analyses. ASCO GI 2024.
[8] ATTRACTION-4: Nivolumab in
gastric cancer (Japanese/Korean population).
[9] KEYNOTE-859: Pembrolizumab
in advanced gastric cancer.
[10] KEYNOTE-062:
Pembrolizumab monotherapy in advanced gastric cancer.